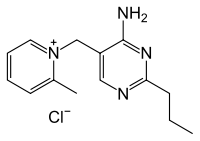
Amprolium
 | |
| Names | |
|---|---|
| IUPAC name
5-[(2-methylpyridin-1-ium-1-yl)methyl]-2-propyl-pyrimidin-4-amine chloride | |
| Other names
1-[(4-Amino-2-propyl-5-pyrimidinyl)methyl]-2-picolinium chloride | |
| Identifiers | |
| 121-25-5 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:85265 |
| ChEMBL | ChEMBL97350 |
| ChemSpider | 66070 |
| ECHA InfoCard | 100.004.054 |
| EC Number | 204-458-4 |
| MeSH | Amprolium |
| PubChem | 2178 |
| UNII | 71M75T660B |
| |
| |
| Properties | |
| C14H19N4+ · Cl− | |
| Molar mass | 278.780 g·mol−1 |
| Pharmacology | |
| QP51AX09 (WHO) | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Amprolium (INN, trade names Amprovine, Amprolium, Amprol, Anticoccid) is a coccidiostat used in poultry.
The drug is a thiamine analogue and blocks the thiamine transporter of Eimeria species. By blocking thiamine uptake it prevents carbohydrate synthesis.
Despite only moderate efficacy it is well favoured due to few resistance issues and is commonly used in conjunction with sulfonamides prophylactically in chickens and cattle as a coccidiostat.
Synthesis
Coccidia are protozoans that can wreak havoc in a flock of poultry by an infection known as coccidiosis. Agents that control this disease–coccidiostats–are in view of the world's heavy dependence on poultry as a source of protein, of great economical significance.
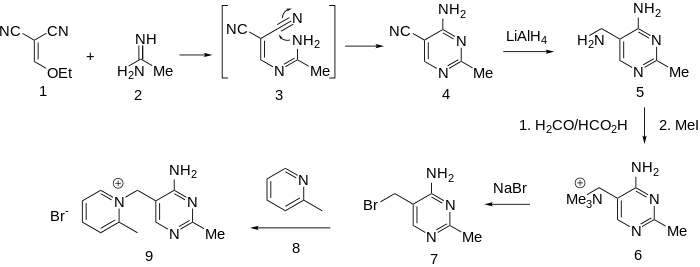
Condensation of ethoxymethylenemalononitrile (1) with acetamidine (2) affords the substituted pyrimidine (4). The rxn may well involve conjugate addition of the amidine nitrogen to the malononitrile followed by loss of ethoxide (3); addition of the remaining amidine nitrogen to one of the nitriles will then lead to the pyrimidine (4). Reduction of the nitrile gives the corresponding aminomethyl compound (5). Exhaustive methylation of the amin followed by displacement of the activated quaternary nitrogen by bromide ion affords the key intermediate (7). Displacement of the halogen by α-picoline gives amprolium.
References
- ↑ Grewe, R. Z. Physiol. Chem. (1936).
- ↑ "Archived copy". Archived from the original on 2015-04-02. Retrieved 2015-03-30.