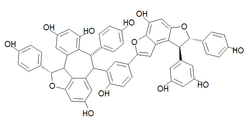
Amurensin K
 | |
| Names | |
|---|---|
| IUPAC name
(1S,6R,7S,11bS)-6-{5-[(7S,8S)-8-(3,5-Dihydroxyphenyl)-4-hydroxy-7-(4-hydroxyphenyl)-7,8-dihydrofuro[3,2-e][1]benzofuran-2-yl]-2-hydroxyphenyl}-1,7-bis(4-hydroxyphenyl)-1,6,7,11b-tetrahydro-2-oxadibenz o[cd,h]azulene-4,8,10-triol | |
| Identifiers | |
| 388121-56-0 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 17288573 |
| PubChem | 16131909 |
| |
| |
| Properties | |
| C56H40O13 | |
| Molar mass | 920.91 g/mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Amurensin K is an oligostilbene. It is a resveratrol tetramer found in Vitis amurensis.[1] Preliminary tests have shown it to be an effective neuraminidase inhibitor against the influenza A virus subtype H1N1.[2]
References
- ↑ Anti-inflammatory tetramers of resveratrol from the roots of Vitis amurensis and the conformations of the seven-membered ring in some oligostilbenes. Kai-Sheng Huang, Mao Lin, and Gui-Fang Cheng, Phytochemistry, September 2001, Volume 58, Issue 2, Pages 357–362, doi:10.1016/S0031-9422(01)00224-2
- ↑ Nguyen, Thi Ngoc Anh; Dao, Trong Tuan; Tung, Bui Thanh; Choi, Hwanwon; Kim, Eunhee; Park, Junsoo; Lim, Seong-IL; Oh, Won Keun (2011). "Influenza A (H1N1) neuraminidase inhibitors from Vitis amurensis". Food Chemistry. 124 (2): 437–443. doi:10.1016/j.foodchem.2010.06.049. ISSN 0308-8146.
This article is issued from Wikipedia - version of the 6/17/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.