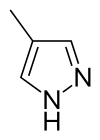
Fomepizole
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
4-Methyl-1H-pyrazole | |||
| Other names
4-Methylpyrazole | |||
| Identifiers | |||
| 7554-65-6 | |||
| 3D model (Jmol) | Interactive image | ||
| ChEBI | CHEBI:5141 | ||
| ChEMBL | ChEMBL1308 | ||
| ChemSpider | 3289 | ||
| DrugBank | DB01213 | ||
| ECHA InfoCard | 100.028.587 | ||
| KEGG | D00707 | ||
| PubChem | 3406 | ||
| UNII | 83LCM6L2BY | ||
| |||
| |||
| Properties | |||
| C4H6N2 | |||
| Molar mass | 82.11 g·mol−1 | ||
| Density | 0.99 g/cm3 | ||
| Boiling point | 204 to 207 °C (399 to 405 °F; 477 to 480 K) (at 97,3 kPa) | ||
| Pharmacology | |||
| V03AB34 (WHO) | |||
| Hazards | |||
| Flash point | 96.0 °C (204.8 °F; 369.1 K) | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Fomepizole or 4-methylpyrazole is indicated for use as an antidote in confirmed or suspected methanol[1] or ethylene glycol[2] poisoning. It may be used alone or in combination with hemodialysis. Apart from medical uses, the role of 4-methylpyrazole in coordination chemistry has been studied.[3]
It is on the World Health Organization's List of Essential Medicines, the most important medications needed in a basic health system.[4]
Medical use
Fomepizole is used in ethylene glycol and methanol toxic ingestion and acts to inhibit the breakdown of these toxins into their active toxic metabolites. Fomepizole is a competitive inhibitor of the enzyme alcohol dehydrogenase,[5] found in the liver. This enzyme plays a key role in the metabolism of ethylene glycol and methanol.
- Ethylene glycol is first metabolized to glycolaldehyde by the enzyme alcohol dehydrogenase, which then undergoes further oxidation to glycolate, glyoxylate, and oxalate. Glycolate and oxalate are primarily responsible for the metabolic acidosis and renal damage seen in ethylene glycol poisoning.
- Methanol is first metabolized to formaldehyde by alcohol dehydrogenase. It then undergoes subsequent oxidation via formaldehyde dehydrogenase to become formic acid.[6] Formic acid is primarily responsible for the metabolic acidosis and visual disturbances associated with methanol poisoning.
By competitively inhibiting the first enzyme in the metabolism of ethylene glycol and methanol, fomepizole slows the production of the toxic metabolites. The slower rate of metabolite production allows the liver to process and excrete the metabolites as they are produced, limiting the accumulation in tissues such as the kidney and eye. As a result, much of the organ damage is avoided.[7]
Fomepizole is most effective when given soon after ingestion of ethylene glycol or methanol. Delaying its administration allows for the generation of harmful metabolites.[7]
Interaction with alcohol
Concurrent use with ethanol is contraindicated because fomepizole is known to prolong the half-life of ethanol. Extending the half-life of ethanol may increase and extend the intoxicating effects of ethanol, allowing for greater (potentially dangerous) levels of intoxication at lower doses. Fomepizole slows the production of acetaldehyde by inhibiting alcohol dehydrogenase, which in turn allows more time to further convert acetaldehyde into acetic acid (vinegar) by acetaldehyde dehydrogenase. The result is a patient with a prolonged and deeper level of intoxication for any given dose of ethanol, and reduced "hangover" symptoms (since these adverse symptoms are largely mediated by acetaldehyde build up). In a chronic alcoholic who has built up a tolerance to ethanol, this removes some of the disincentives to ethanol consumption ("negative reinforcement") while allowing them to become intoxicated with a lower dose of ethanol. The danger is that the alcoholic will then overdose on ethanol (possibly fatally). If alcoholics instead very carefully reduce their doses to reflect the now slower metabolism, they may get the "rewarding" stimulus of intoxication at lower doses with less adverse "hangover" effects - leading potentially to increased psychological dependency. However, these lower doses may therefore produce less chronic toxicity and provide a harm minimization approach to chronic alcoholism. It is, in essence, the antithesis of a disulfiram approach which tries to increase the buildup of acetaldehyde resulting in positive punishment for the patient (needless to say compliance / adherence is a substantial problem in disulfiram-based approaches). Disulfiram also has a considerably longer half-life than that of fomepizole, requiring the person to not drink ethanol in order to avoid severe effects. If the person is not adequately managed on a benzodiazepine, barbiturate, acamprosate, or another GABAA receptor agonist, the alcohol withdrawal syndrome (known as delerium tremens "DT") may occur; disulfiram treatment should never be initiated until the risk for DT has been evaluated and mitigated appropriately, but fomepazole treatment may be initiated while the DT de-titration sequence is still being calibrated based upon person's withdrawal symptoms and psychological health.
Adverse effects
Common side effects associated with fomepizole use include headache and nausea.[8]
Kinetics
Absorption and distribution
Fomepizole distributes rapidly into total body water. The volume of distribution is between 0.6 and 1.02 l/kg. The therapeutic concentration is from 8.2 to 24.6 mg (100 to 300 micromoles) per liter. Peak concentration following single oral doses of 7 to 50 mg/kg of body weight occurred in 1 to 2 hours. The half-life varies with dose, so has not been calculated.
Metabolism and elimination
Hepatic; the primary metabolite is 4-carboxypyrazole (about 80 to 85% of an administered dose). Other metabolites include the pyrazoles 4-hydroxymethylpyrazole and the N -glucuronide conjugates of 4-carboxypyrazole and 4-hydroxymethylpyrazole.
Following multiple doses, fomepizole rapidly induces its own metabolism via the cytochrome P450 mixed-function oxidase system.
In healthy volunteers, 1.0 to 3.5% of an administered dose was excreted unchanged in the urine. The metabolites also are excreted unchanged in the urine.
Fomepizole is dialyzable.
References
- ↑ International Programme on Chemical Safety (IPCS): Methanol (PIM 335), , retrieved on March 1, 2008
- ↑ Velez LI, Shepherd G, Lee YC, Keyes DC (September 2007). "Ethylene glycol ingestion treated only with fomepizole" (PDF). J Med Toxicol. 3 (3): 125–8. doi:10.1007/BF03160922. PMID 18072148.
- ↑ Vos, Johannes G.; Groeneveld, Willem L. (1979). "Pyrazolato and related anions. Part V. Transition metal salts of 4-methylpyrazole". Transition Metal Chemistry. 4 (3): 137. doi:10.1007/BF00619054.
- ↑ "WHO Model List of EssentialMedicines" (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- ↑ Casavant MJ (January 2001). "Fomepizole in the treatment of poisoning". Pediatrics. 107 (1): 170. doi:10.1542/peds.107.1.170. PMID 11134450.
- ↑ "Forensic Pathology".
- 1 2 "Fomepizole for Ethylene Glycol and Methanol Poisoning".
- ↑ Lepik, KJ; Levy, AR; Sobolev, BG; Purssell, RA; DeWitt, CR; Erhardt, GD; Kennedy, JR; Daws, DE; Brignall, JL (Apr 2009). "Adverse drug events associated with the antidotes for methanol and ethylene glycol poisoning: a comparison of ethanol and fomepizole.". Annals of Emergency Medicine. 53 (4): 439–450.e10. doi:10.1016/j.annemergmed.2008.05.008. PMID 18639955.