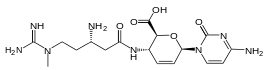
Blasticidin S
 | |
| Names | |
|---|---|
| IUPAC name
4-amino-1-[4-({(3S)-3-amino-5-[[amino(imino)methyl](methyl)amino]pentanoyl}amino)-2,3,4-trideoxy-β-D-erythro-hex-2-enopyranuronosyl]pyrimidin-2(1H)-one | |
| Identifiers | |
| 2079-00-7 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:15353 |
| ChEMBL | ChEMBL476894 |
| ChemSpider | 148673 |
| ECHA InfoCard | 100.109.057 |
| KEGG | C02010 |
| PubChem | 258 |
| |
| |
| Properties | |
| C17H26N8O5 | |
| Molar mass | 422.44 g/mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Blasticidin S is an antibiotic that is produced by Streptomyces griseochromogenes. In biological research, specifically genetic engineering, it is used to select transformed cells which have been engineered to carry a resistance gene for blasticidin. In short, DNA of interest is fused to DNA encoding a resistance gene, and then is transformed into cells. After allowing time for recovery and for cells to begin transcribing and translating their new DNA, blasticidin is added. Now only the cells that have the new DNA can grow.
History
In the 1950s, a drug screening program was designed in Japan to discover a new antibiotic that prevents Blast Disease by the fungus Magnaporthe grisea.[1]
Resistance genes
Three resistance genes have been cloned:
- bls (an acetyltransferase) from Streptoverticillum sp. which itself produces blasticidin in a natural example of biological warfare
- bsr (a blasticidin-S deaminase) from Bacillus cereus (other bsr genes are known as well, see listings in Genbank)
- BSD (another deaminase) from Aspergillus terreus
bsr and BSD are the most commonly used resistance genes. The proteins produced from these genes enable the cells carrying them to produce protein in the presence of blasticidin.
Mechanism of action
Blasticidin prevents the growth of both eukaryotic and prokaryotic cells. It works by inhibiting termination step of translation and peptide bond formation (to lesser extent) by the ribosome. This means that cells can no longer produce new proteins through translation of mRNA.
References
- ↑ Natural Products Isolation: Separation Methods for Antimicrobials, Antivirals, and Enzyme Inhibitors. Wagman G. H., Elsevier R. C.; p. 191 (1988).