Brassinosteroid
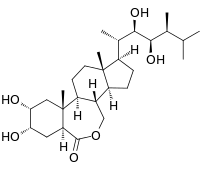
Brassinosteroids (BRs) are a class of polyhydroxysteroids that have been recognized as a sixth class of plant hormones. These were first explored nearly 40 years ago, when Mitchell et al. reported promotion in stem elongation and cell division by the treatment of organic extracts of rapeseed (Brassica napus) pollen.[1] Brassinolide was the first isolated brassinosteroid in 1979, when pollen from Brassica napus was shown to promote stem elongation and cell divisions, and the biologically active molecule was isolated.[1][2] The yield of brassinosteroids from 230 kg of Brassica napus pollen was only 10 mg. Since their discovery, over 70 BR compounds have been isolated from plants.[3]
Biosynthesis
The BR is biosynthesised from campesterol. The biosynthetic pathway was elucidated by Japanese researchers and later shown to be correct through the analysis of BR biosynthesis mutants in Arabidopsis thaliana, tomatoes, and peas.[4] The sites for BR synthesis in plants have not been experimentally demonstrated. One well-supported hypothesis is that all tissues produce BRs, since BR biosynthetic and signal transduction genes are expressed in a wide range of plant organs, and short distance activity of the hormones also supports this.[5][6] Experiments have shown that long distance transport is possible and that flow is in an acropetal direction, but it is not known if this movement is biologically relevant.[5] Brassinosteroids are recognized at the cell membrane, although they are membrane-soluble.
Hormonal activity
BRs have been shown to be involved in numerous plant processes:
- Promotion of cell expansion and cell elongation;[5] works with auxin to do so.[7]
- It has an unclear role in cell division and cell wall regeneration.[5]
- Promotion of vascular differentiation; BR signal transduction has been studied during vascular differentiation.[8]
- Is necessary for pollen elongation for pollen tube formation.[9]
- Acceleration of senescence in dying tissue cultured cells; delayed senescence in BR mutants supports that this action may be biologically relevant.[5]
- Can provide some protection to plants during chilling and drought stress.[5]
Extract from the plant Lychnis viscaria contains a relatively high amount of Brassinosteroids. Lychnis viscaria increases the disease resistance of surrounding plants.
24-Epibrassinolide (EBL), a brassinosteroid isolated from Aegle marmelos Correa (Rutaceae), was further evaluated for the antigenotoxicity against maleic hydrazide (MH)-induced genotoxicity in Allium cepa chromosomal aberration assay. It was shown that the percentage of chromosomal aberrations induced by maleic hydrazide (0.01%) declined significantly with 24-epibrassinolide treatment.[10]
BRs have been reported to counteract both abiotic and biotic stress in plants.[11][12] Application of brassinosteroids to cucumbers was demonstrated to increase the metabolism and removal of pesticides, which could be beneficial for reducing the human ingestion of residual pesticides from non-organically grown vegetables.[13] In all Type of brassinosteroids 28-homoBL is the most effective type of brassinosteroids. (sandeep kumar et al. 2010 Jour. of Indian bot society) Brassinosteroids increased tolerance to high temperature in Brassica juncea L. (Kumar S. 2010) The ability of 28-homobrassinolide to confer resistance to stress in Brassica juncea L. has also established (sandeep kumar). Application of 24-epiBL have any protective role on shoot, root length, soluble protein, proline content and peroxidases along with proline content PPO and IAA in seedlings of B. juncea L. under seasonal stress (Geetika Sirhindi)
BRs have also been reported to have a variety of effects when applied to rice seeds (Oryza sativa L.).Seeds treated with BRs were shown to reduce the growth inhibitory effect of salt stress.[14] When the developed plants fresh weight was analyzed the treated seeds outperformed plants grown on saline and non-saline medium however when the dry weight was analyzed BR treated seeds only outperformed untreated plants that were grown on saline medium.[14] When dealing with tomatoes (Lycopersicon esculentum) under salt stress the concentration of cholophyll a and cholophyll b were decreased and thus pigmentation was decreased as well.[15] BR treated rice seeds considerably restored the pigment level in plants that were grown on saline medium when compared to non-treated plants under the same conditions.[14]
Agricultural uses
BR might reveal to have a prominent interest in the role of horticultural crops. Based on extensive research BR has the ability to improve the quantity and quality of horticultural crops and protect plants against many stresses that can be present in the local environment.[16][17] With the many advances in technology dealing with the synthesis of more stable synthetic analogues and the genetic manipulation of cellular BR activity, using BR in the production of horticultural crops has become a more practical and hopeful strategy for improving crop yields and success.[16]
BR could also help bridge the gap of the consumers' health concerns and the producers need for growth. A major benefit of using BR is that it does not interfere with the environment because they act in natural doses in a natural way.[17] Since it is a “plant strengthening substance” and it is natural, BR application would be more favorable than pesticides and does not contribute to the co-evolution of pests.[17]
In Germany, extract from the plant is allowed for use as a "plant strengthening substance."
Detection and chemical analysis
BRs can be detected by gas chromatography mass spectrometry and bioassays.[18]
References
- 1 2 Grove, Michael D.; Spencer, Gayland F.; Rohwedder, William K.; Mandava, Nagabhushanam; Worley, Joseph F.; Warthen, J. David; Steffens, George L.; Flippen-Anderson, Judith L.; Cook, J. Carter (1979). "Brassinolide, a plant growth-promoting steroid isolated from Brassica napus pollen". Nature. 281 (5728): 216–217. Bibcode:1979Natur.281..216G. doi:10.1038/281216a0.
- ↑ Grove, M.D.; Spencer, G.F.; Rohwedder, W.K.; Mandava, N.; Worley, J.F.; Warthen, J.D.; Ste; Flippen-Anderson, J.L.; Cook, J.C.; et al. (1979). "Brassins: a new family of plant hormones from rape pollen". Nature. 225 (5237): 1065–66. Bibcode:1970Natur.225.1065M. doi:10.1038/2251065a0. PMID 16056912.
- ↑ Bajguz, A. (2007). "Metabolism of brassinosteroids in plants". Plant Physiology and Biochemistry. 45 (2): 95–107. doi:10.1016/j.plaphy.2007.01.002. PMID 17346983.
- ↑ Fujioka, S; Sakurai, A. (1997). "Biosynthesis and metabolism of brassinosteroids". Physiologia Plantarum. 100 (3): 710–15. doi:10.1111/j.1399-3054.1997.tb03078.x.
- 1 2 3 4 5 6 Clouse, SD; Sasse, JM. (1998). "Brassinosteroids: Essential regulators of plant growth and development". Annu. Rev. Plant Physiol. Plant Mol. Biol. 49: 427–51. doi:10.1146/annurev.arplant.49.1.427.
- ↑ Li, JM; Chory, J. (1997). "A putative leucine rich repeat receptor kinase involved in brassinosteroid signal transduction". Cell. 90 (5): 929–38. doi:10.1016/S0092-8674(00)80357-8. PMID 9298904.
- ↑ Nemhauser, Jennifer L.; Mockler, Todd C.; Chory, Joanne (2004). "Interdependency of Brassinosteroid and Auxin Signaling in Arabidopsis". PLoS Biology. 2 (9): e258. doi:10.1371/journal.pbio.0020258. PMC 509407
 . PMID 15328536.
. PMID 15328536. - ↑ Caño-Delgado, A; Yin, Y; Yu, C; Vafeados, D; Mora-Garcia, S; Cheng, JC; Nam, KH; Li, J; Chory, J (2004). "BRL1 and BRL3 are novel brassinosteroid receptors that function in vascular differentiation in Arabidopsis". Development (Cambridge, England). 131 (21): 5341–51. doi:10.1242/dev.01403. PMID 15486337.
- ↑ Hewitt, FR; Hough, T; O'Neill, P; Sasse, JM; Williams, EG; Rowan, KS (1985). "Effect of brassinolide and other growth regulators on the germination and growth of pollen tubes of "Prunus avium" using a multiple hanging drop assay". Aust. J Plant Physiol. 12 (2): 201–11. doi:10.1071/PP9850201.
- ↑ Sondhi, N.; Bhardwaj, R.; Kaur, S.; Singh, B.; Kumar, N. (2008). "Isolation of 24-epibrassinolide from leaves of "Aegle marmelos" and evaluation of its antigenotoxicity potential employing Allium cepa chromosomal aberration assay". Plant Growth Regul. 54 (3): 217–224. doi:10.1007/s10725-007-9242-7.
- ↑ Sharma, P.; Bhardwaj, R. (2007). "Effects of 24-Epibrassinolide on growth and metal uptake in "Brassica juncea" L. under copper metal stress". Acta Physiologiae Plantarum. 29 (3): 259–263. doi:10.1007/s11738-007-0032-7.
- ↑ Sharma, P; Bhardwaj, R; Arora, HK; Arora, N; Kumar, A. (2008). "Effects of 28-homobrassinolide on nickel uptake, protein content and antioxidative defence system in "Brassica juncea". Biol. Plant. 52 (4): 767–770. doi:10.1007/s10535-008-0149-6.
- ↑ Xiao Jian, Xia; Zhang, Y; Wu, JX; Wang, JT; Zhou, YH; Shi, K; Yu, YL; Yu, JQ (2009). "Brassinosteroids Promote Metabolism of Pesticides in Cucumber". J. Agric. Food Chem. 57 (18): 8406–8413. doi:10.1021/jf901915a. PMID 19694443.
- 1 2 3 Anuradha, S, and S Seeta Ram Rao. Application of brassinosteroids to rice seeds (Oryza sativa L.) reduced the impact of salt stress on growth, prevented photosynthetic pigment loss and increased nitrate reductase activity. Vol. 40. N.p.: Plant Growth Regulation, 2003. 29-32. Print.
- ↑ El-Meleigy, El-Sayed A., Mahdi F. Gabr, Fouad H. Mohamed, and Mona A. Ismail. Responses to NaCl Salinity of Tomato Cultivated and Breeding Lines Differing in Salt Tolerance in Callus Cultures. N.p.: INTERNATIONAL JOURNAL OF AGRICULTURE & BIOLOGY, 2004. 1560-8530. Print.
- 1 2 Kang, Y Y., and S R. Guo. ROLE OF BRASSINOSTEROIDS ON HORTICULTURAL CROPS. N.p.: College of Horticulture, n.d. 269-88. Print.
- 1 2 3 Khripach, VLADIMIR, Vlandimir Zhabinsk II, and Aede D. Groot. Twenty Years of Brassinosteroids: Steroidal Plant Hormones Warrant Better Crops for the XXI Century. 86th ed. N.p.: Annals of Botany, 2000. 441-47. Print.
- ↑ Kim, Seong-Ki, Hiroshi Abe, C. H. Anthony Little, and Richard P. Pharis. Identification of Two Brassinosteroids from the Cambial Region of Scots Pine (Pinus silverstris) by Gas Chromatography-Mass Spectrometry, after Detection Using a Dwarf Rice Lamina Inclination Bioassay. N.p.: Plant Physiology, 1990. 1709-13. Print.