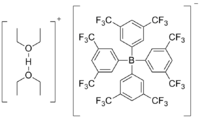
Brookhart's acid
 | |
| Identifiers | |
|---|---|
| 139362-04-2 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 9301081 |
| PubChem | 11125959 |
| |
| |
| Properties | |
| C40H33BF24O2 | |
| Molar mass | 1012.46 |
| Appearance | White crystalline solid |
| Hazards | |
| Main hazards | Strong acid |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Brookhart's acid is the salt of the diethyl ether oxonium ion and tetrakis[3,5-bis(trifluoromethyl)phenyl]borate (BAr’4). It is a colorless solid, used as a strong acid. The compound was first reported by Volpe, Grant, and Brookhart in 1992.[1]
Preparation
This compound is prepared by treatment of NaBAr’4 in diethyl ether (Et2O) with hydrogen chloride:
- NaBAr'4 + HCl + 2 Et2O → [H(OEt2)2]+BAr'4− + NaCl
NaBAr’4 is soluble in diethyl ether, whereas sodium chloride is not. Precipitation of sodium chloride thus drives the formation of the oxonium acid compound, which is isolable as a solid.[1]
Structure and properties
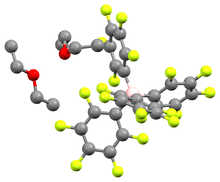
The acid crystallizes as a white, hygroscopic crystalline solid. NMR and elemental analysis showed that the crystal contains two equivalents of diethyl ether. In solution, the compound slowly degrades to m-C6H3(CF3)2 and BAr'3.[1]
[H(OEt2)2][B(C6F5)4] is a related compound with a slightly different weakly coordinating anion; it was first reported in 2000. An X-ray crystal structure of that compound was obtained, showing the acidic proton coordinated by both ethereal oxygen centers, although the crystal was not good enough to determine whether the proton is located symmetrically or unsymmetrically between the two.[2]
Uses
Traditional weakly coordinating anions, such as perchlorate, tetrafluoroborate, and hexafluorophosphate will coordinate to very electrophilic cations, making these counterions unsuitable for some complexes.[3] The highly reactive species [Cp2Zr(CH3)]+, for example, has been reported to abstract F− from PF6.[4] Starting in the 1980s, new types weakly coordinating anions began to be developed. BAr’4 anions are used as counterions for highly electrophilic, cationic transition metal species, as they are very weakly coordinating and unreactive towards electrophilic attack.[1] One common method of generating these cationic species is via protonolysis of a dialkyl complexes or an olefin complex. For example, an electrophilic palladium catalyst, [(2,2'-bipyridine)Pd(CH3)(CH3CN)][BAr'4], is prepared by protonating the dimethyl complex with Brookhart's acid. This electrophilic, cationic palladium species is used for the polymerization of olefins with carbon monoxide to polyketones in aprotic solvents.[5]
References
- 1 2 3 4 Brookhart, M.; Grant, B.; Volpe, A. F. (1992). "[(3,5-(CF3)2C6H3)4B]−[H(OEt2)2]+: A convenient reagent for generation and stabilization of cationic, highly electrophilic organometallic complexes". Organometallics. 11 (11): 3920. doi:10.1021/om00059a071.
- 1 2 Jutzi, P.; Müller, C.; Stammler, A.; Stammler, H. G. (2000). "Synthesis, Crystal Structure, and Application of the Oxonium Acid [H(OEt2)2]+B(C6F5)4]−". Organometallics vol. 19, p. 1442. doi:10.1021/om990612w
- ↑ Krossing, I.; Raabe, I. (2004). "Noncoordinating Anions—Fact or Fiction? A Survey of Likely Candidates". Angewandte Chemie International Edition. 43 (16): 2066–90. doi:10.1002/anie.200300620. PMID 15083452.
- ↑ Jordan, R. F.; Dasher, W. E.; Echols, S. F. (1986). "Reactive cationic dicyclopentadienyl zirconium(IV) complexes". Journal of the American Chemical Society. 108 (7): 1718. doi:10.1021/ja00267a068.
- ↑ Brookhart, M.; Rix, F. C.; Desimone, J. M.; Barborak, J. C. (1992). "Palladium(II) catalysts for living alternating copolymerization of olefins and carbon monoxide". Journal of the American Chemical Society. 114 (14): 5894. doi:10.1021/ja00040a082.