CPK coloring
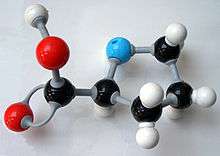
In chemistry, the CPK coloring is a popular color convention for distinguishing atoms of different chemical elements in molecular models. The scheme is named after the CPK molecular models designed by chemists Robert Corey and Linus Pauling, and improved by Walter Koltun.
History
In 1952, Corey and Pauling published a description of space-filling models of proteins and other biomolecules that they had been building at Caltech.[1] Their models represented atoms by faceted hardwood balls, painted in different bright colors to indicate the respective chemical elements. Their color schema included
They also built smaller models using plastic balls with the same color schema.
In 1965 Koltun patented an improved version of the Corey and Pauling modeling technique.[2] In his patent he mentions the following colors:
- White for hydrogen
- Black for carbon
- Blue for nitrogen
- Red for oxygen
- Deep yellow for sulfur
- Purple for phosphorus
- Light, medium, medium dark, and dark green for the halogens (F, Cl, Br, I)
- Silver for metals (Co, Fe, Ni, Cu)
Typical assignments

Typical CPK color assignments include:
| hydrogen (H) | white | |
| carbon (C) | black | |
| nitrogen (N) | dark blue | |
| oxygen (O) | red | |
| fluorine (F), chlorine (Cl) | green | |
| bromine (Br) | dark red | |
| iodine (I) | dark violet | |
| noble gases (He, Ne, Ar, Xe, Kr) | cyan | |
| phosphorus (P) | orange | |
| sulfur (S) | yellow | |
| boron (B), most transition metals | peach, salmon | |
| alkali metals (Li, Na, K, Rb, Cs, Fr) | violet | |
| alkaline earth metals (Be, Mg, Ca, Sr, Ba, Ra) | dark green | |
| titanium (Ti) | gray | |
| iron (Fe) | orange | |
| other elements | pink |
Several of the CPK colors refer mnemonically to colors of the pure elements or notable compound. For example, hydrogen is a colorless gas, carbon as charcoal or graphite is black, common sulfur is yellow, chlorine is a greenish gas, bromine is a dark red liquid, iodine in ether is violet, amorphous phosphorus is red, rust is dark orange-red, etc. For some colors, such as those of oxygen and nitrogen, the inspiration is less clear. Perhaps red for oxygen is inspired by the fact that oxygen is normally required for combustion or that the oxygen-bearing chemical in blood, hemoglobin, is bright red, and the blue for nitrogen by the fact that nitrogen is the main component of Earth's atmosphere, which appears to human eyes as being colored sky blue.
Modern variants
The following table shows colors assigned to each element by some popular software products. Column C is the original assignment by Corey and Pauling,[1] and K is that of Koltun's patent.[2] Column J is the color scheme used by the molecular visualizer Jmol.[3] Column R is the scheme used by Rasmol; when two colors are shown, the second one is valid for versions 2.7.3 and later.[3][4] All colors are approximate and may depend on the display hardware and viewing conditions.
See also
References
- 1 2 Robert B. Corey and Linus Pauling (1953): Molecular Models of Amino Acids, Peptides, and Proteins. Review of Scientific Instruments, Volume 24, Issue 8, pp. 621-627. doi:10.1063/1.1770803
- 1 2 Walter L. Koltun (1965), Space filling atomic units and connectors for molecular models. U. S. Patent 3170246.
- 1 2 Jmol color table at sourceforge.net. Accessed on 2010-01-28.
- ↑ Rasmol color table at bio.cmu.edu. Accessed on 2010-01-28.
External links
- What is the basis for the CPK color scheme? Which colors is used for which atom?
- Physical Molecular Models