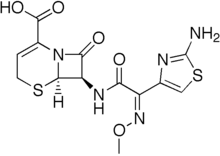
Ceftizoxime
 | |
| Clinical data | |
|---|---|
| Trade names | Cefizox |
| AHFS/Drugs.com | Consumer Drug Information |
| MedlinePlus | a684043 |
| ATC code | J01DD07 (WHO) |
| Identifiers | |
| |
| CAS Number | 68401-81-0 68401-82-1 |
| PubChem (CID) | 6533629 |
| ChemSpider | 5018818 |
| UNII |
C43C467DPE |
| KEGG | D07658 |
| ChEMBL | CHEMBL528 |
| ECHA InfoCard | 100.210.846 |
| Chemical and physical data | |
| Formula | C13H13N5O5S2 |
| Molar mass | 383.405 g/mol |
Ceftizoxime is a third-generation cephalosporin available for parenteral administration. Unlike other third-generation cephalosporins, the whole C-3 side chain in ceftizoxime has been removed to prevent deactivation by hydrolytic enzymes. It rather resembles cefotaxime in its properties, but is not subject to metabolism. It was removed from the US Market in 2007.
Synthesis
Injectable third generation cephalosporin antibiotic related to cefotaxime, q.v. Exhibits broad spectrum activity and resistance to β-lactamase hydrolysis.
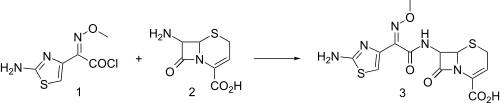
Ceftizoxime synthesis:[1]
References
- ↑ T. Takaya et al., DE 2810922; eidem, U.S. Patent 4,427,674 (1978, 1984 both to Fujisawa).
This article is issued from Wikipedia - version of the 3/15/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.