Dermcidin
Dermcidin, also known as proteolysis-inducing factor (PIF), is a protein that in humans is encoded by the DCD gene.[2][3] It is an anti-microbial peptide[3] secreted by human eccrine sweat glands onto the skin as a part of the innate host defense of the immune system. It is also involved in proteolysis.[3]
Function
Dermcidin is a secreted protein that is subsequently processed into mature peptides of distinct biological activities. The C-terminal peptide is constitutively expressed in sweat and has antibacterial and antifungal activities. The N-terminal peptide, also known as diffusible survival evasion peptide, promotes neural cell survival under conditions of severe oxidative stress. A glycosylated form of the N-terminal peptide may be associated with cachexia (muscle wasting) in cancer patients.[3]
Survival evasion peptide Antimicrobial peptide
YDPEAASAPGSGNPCHEASAAQKENAGEDPGLARQAPKPRKQRSSLLEKGLDGAKKAVGGLGKLGKDAVEDLESVGKGAVHDVKDVLDSVL
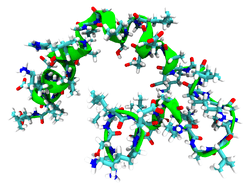
The C-termial precursor DCD-1L is a 48 residue peptide that shows partial helicity in solution, as evidenced by the determination of its solution structure by NMR and CD-spectroscopy. The full length precursor is processed by undetermined proteases present in human sweat, to form several shorter peptides that show variable antimicrobial activity, named according to their C-terminal triplet of amino acids and their resiude length. One such active peptide is SSL25, which shows a 2-fold increase in activity against E.coli compared to DCD-1L.[4]
DCD-1L SSLLEKGLDGAKKAVGGLGKLGKDAVEDLESVGKGAVHDVKDVLDSVL
DCD-1 SSLLEKGLDGAKKAVGGLGKLGKDAVEDLESVGKGAVHDVKDVLDSV
SSL25 SSLLEKGLDGAKKAVGGLGKLGKDA
Mechanism
The crystal structure of dermcidin has been solved in solution to reveal a hexameric helix-bundle, mediated by Zn ion binding.[5] This is observed to form a tilted channel in membranes under computational examination by molecular dynamics simulations, and one suggested mechanism of antimicrobial action inferred from this observation is by ion gradient decoupling across biological membranes. This is supported by concurrent observations in experimental studies of a voltage dependent depolarization of lipid bilayers.
References
- ↑ "Human PubMed Reference:".
- ↑ Schittek B, Hipfel R, Sauer B, Bauer J, Kalbacher H, Stevanovic S, Schirle M, Schroeder K, Blin N, Meier F, Rassner G, Garbe C (Nov 2001). "Dermicidin: a novel human antibiotic peptide secreted by sweat glands". Nat Immunol. 2 (12): 1133–7. doi:10.1038/ni732. PMID 11694882.
- 1 2 3 4 "Entrez Gene: DCD dermcidin".
- ↑ Baechle D, Flad T, Cansier A, et al. (2006). "Cathepsin D is present in human eccrine sweat and involved in the postsecretory processing of the antimicrobial peptide DCD-1L.". J. Biol. Chem. 281 (9): 5406–15. doi:10.1074/jbc.M504670200. PMID 16354654.
- ↑ PDB: 2YMKSong, Chen; Weichbrodt, Conrad; Salnikov, Evgeniy S; Dynowski, Marek; Forsberg, Björn O; Bechinger, Burkhard; Steinem, Claudia; de Groot, Bert L; Zachariae, Ulrich; Zeth, Kornelius (Feb 2013). "Crystal structure and functional mechanism of a human antimicrobial membrane channel". PNAS. 110 (12): 4586–91. doi:10.1073/pnas.1214739110. PMID 23426625.
Further reading
- Todorov P, Cariuk P, McDevitt T, et al. (1996). "Characterization of a cancer cachectic factor.". Nature. 379 (6567): 739–42. doi:10.1038/379739a0. PMID 8602222.
- Todorov PT, Deacon M, Tisdale MJ (1997). "Structural analysis of a tumor-produced sulfated glycoprotein capable of initiating muscle protein degradation.". J. Biol. Chem. 272 (19): 12279–88. doi:10.1074/jbc.272.19.12279. PMID 9139670.
- Cunningham TJ, Hodge L, Speicher D, et al. (1998). "Identification of a survival-promoting peptide in medium conditioned by oxidatively stressed cell lines of nervous system origin.". J. Neurosci. 18 (18): 7047–60. PMID 9736629.
- Cunningham TJ, Jing H, Wang Y, Hodge L (2000). "Calreticulin binding and other biological activities of survival peptide Y-P30 including effects of systemic treatment of rats.". Exp. Neurol. 163 (2): 457–68. doi:10.1006/exnr.2000.7390. PMID 10833321.
- Cunningham TJ, Jing H, Akerblom I, et al. (2002). "Identification of the human cDNA for new survival/evasion peptide (DSEP): studies in vitro and in vivo of overexpression by neural cells.". Exp. Neurol. 177 (1): 32–9. doi:10.1006/exnr.2002.7979. PMID 12429208.
- Strausberg RL, Feingold EA, Grouse LH, et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences.". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. doi:10.1073/pnas.242603899. PMC 139241
 . PMID 12477932.
. PMID 12477932.
- Porter D, Weremowicz S, Chin K, et al. (2003). "A neural survival factor is a candidate oncogene in breast cancer.". Proc. Natl. Acad. Sci. U.S.A. 100 (19): 10931–6. doi:10.1073/pnas.1932980100. PMC 196905
 . PMID 12953101.
. PMID 12953101.
- Zhang Z, Henzel WJ (2005). "Signal peptide prediction based on analysis of experimentally verified cleavage sites.". Protein Sci. 13 (10): 2819–24. doi:10.1110/ps.04682504. PMC 2286551
 . PMID 15340161.
. PMID 15340161.
- Suzuki Y, Yamashita R, Shirota M, et al. (2004). "Sequence comparison of human and mouse genes reveals a homologous block structure in the promoter regions.". Genome Res. 14 (9): 1711–8. doi:10.1101/gr.2435604. PMC 515316
 . PMID 15342556.
. PMID 15342556.
- Monitto CL, Dong SM, Jen J, Sidransky D (2005). "Characterization of a human homologue of proteolysis-inducing factor and its role in cancer cachexia.". Clin. Cancer Res. 10 (17): 5862–9. doi:10.1158/1078-0432.CCR-04-0435. PMID 15355918.
- Gerhard DS, Wagner L, Feingold EA, et al. (2004). "The status, quality, and expansion of the NIH full-length cDNA project: the Mammalian Gene Collection (MGC).". Genome Res. 14 (10B): 2121–7. doi:10.1101/gr.2596504. PMC 528928
 . PMID 15489334.
. PMID 15489334.
- Lai YP, Peng YF, Zuo Y, et al. (2005). "Functional and structural characterization of recombinant dermcidin-1L, a human antimicrobial peptide.". Biochem. Biophys. Res. Commun. 328 (1): 243–50. doi:10.1016/j.bbrc.2004.12.143. PMID 15670776.
- Rieg S, Steffen H, Seeber S, et al. (2005). "Deficiency of dermcidin-derived antimicrobial peptides in sweat of patients with atopic dermatitis correlates with an impaired innate defense of human skin in vivo.". J. Immunol. 174 (12): 8003–10. doi:10.4049/jimmunol.174.12.8003. PMID 15944307.
- Watchorn TM, Dowidar N, Dejong CH, et al. (2006). "The cachectic mediator proteolysis inducing factor activates NF-kappaB and STAT3 in human Kupffer cells and monocytes.". Int. J. Oncol. 27 (4): 1105–11. doi:10.3892/ijo.27.4.1105. PMID 16142329.
- Lowrie AG, Wigmore SJ, Wright DJ, et al. (2006). "Dermcidin expression in hepatic cells improves survival without N-glycosylation, but requires asparagine residues.". Br. J. Cancer. 94 (11): 1663–71. doi:10.1038/sj.bjc.6603148. PMC 2361319
 . PMID 16685272.
. PMID 16685272.
- Ewing RM, Chu P, Elisma F, et al. (2007). "Large-scale mapping of human protein-protein interactions by mass spectrometry.". Mol. Syst. Biol. 3 (1): 89. doi:10.1038/msb4100134. PMC 1847948
 . PMID 17353931.
. PMID 17353931.
- Frum R, Busby SA, Ramamoorthy M, et al. (2007). "HDM2-binding partners: interaction with translation elongation factor EF1alpha.". J. Proteome Res. 6 (4): 1410–7. doi:10.1021/pr060584p. PMID 17373842.
- Lee Motoyama JP, Kim-Motoyama H, Kim P, et al. (2007). "Identification of dermcidin in human gestational tissue and characterization of its proteolytic activity.". Biochem. Biophys. Res. Commun. 357 (4): 828–33. doi:10.1016/j.bbrc.2007.03.112. PMID 17448443.
External links
 . PMID 12477932.
. PMID 12477932. . PMID 12953101.
. PMID 12953101. . PMID 15340161.
. PMID 15340161. . PMID 15342556.
. PMID 15342556. . PMID 15489334.
. PMID 15489334. . PMID 16685272.
. PMID 16685272. . PMID 17353931.
. PMID 17353931.