Diphosphorus tetraiodide
 | |
| Names | |
|---|---|
| IUPAC name
Diphosphorus tetraiodide | |
| Preferred IUPAC name
Tetraiododiphosphane | |
| Other names
Phosphorus(II) iodide | |
| Identifiers | |
| 13455-00-0 | |
| ECHA InfoCard | 100.033.301 |
| Properties | |
| P2I4 | |
| Molar mass | 569.57 g/mol |
| Appearance | Orange crystalline solid |
| Melting point | 124 to 127 °C (255 to 261 °F; 397 to 400 K) |
| Boiling point | Decomposes |
| Decomposes | |
| Hazards | |
| EU classification (DSD) |
|
| R-phrases | R14, R34, R37 |
| Flash point | Non-flammable |
| Related compounds | |
| Other anions |
Diphosphorus tetrafluoride Diphosphorus tetrachloride Diphosphorus tetrabromide |
| Other cations |
diarsenic tetraiodide |
| Related Binary Phosphorus halides |
phosphorus triiodide |
| Related compounds |
diphosphane diphosphines |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Diphosphorus tetraiodide is an orange crystalline solid with the formula P2I4. It has been used as a reducing agent in organic chemistry. It is a rare example of a compound with phosphorus in the +2 oxidation state, and can be classified as a subhalide of phosphorus. It is the most stable of the diphosphorus tetrahalides.[1]
Synthesis and structure
Diphosphorus tetraiodide is easily generated by the disproportionation of phosphorus triiodide in dry ether:
- 2 PI3 → P2I4 + I2
It can also be obtained by treating phosphorus trichloride and potassium iodide in anhydrous conditions.[2]
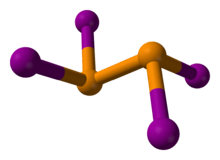
The compound adopts a centrosymmetric structure with a P-P bond of 2.230 Å.[3]
Reactions
Inorganic chemistry
Diphosphorus tetraiodide reacts with bromine to form mixtures PI3-xBrx. With sulfur, it is oxidized to P2S2I4, retaining the P-P bond.[1]
Organic chemistry
Diphosphorus tetraiodide is used in organic synthesis mainly as a deoxygenating agent.[4] It is used for deprotecting acetals and ketals to aldehydes and ketones, and for converting epoxides into alkenes and aldoximes into nitriles. It can also cyclize 2-aminoalcohols to aziridines[5] and to convert α,β-unsaturated carboxylic acids to α,β-unsaturated bromides.[6]
As foreshadowed by the work of Bertholet in 1855,[4] diphosphorus tetraiodide is used in the Kuhn–Winterstein reaction, the conversion of glycols to alkenes.[7]
References
- 1 2 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9.
- ↑ H. Suzuki; T. Fuchita; A. Iwasa; T. Mishina (December 1978). "Diphosphorus Tetraiodide as a Reagent for Converting Epoxides into Olefins, and Aldoximes into Nitriles under Mild Conditions". Synthesis. 1978 (12): 905–908. doi:10.1055/s-1978-24936.
- ↑ Z. Žák, M. Černík "Diphosphorus tetraiodide at 120 K" Acta Crystallographica, Section C: Crystal Structure Communications 1996, vol. C52, pp. 290-1. doi:10.1107/S0108270195012510
- 1 2 Alain Krief, Vikas N. Telvekar "Diphosphorus Tetraiodide" Encyclopedia for Reagents in Organic Synthesis 2009. doi:10.1002/047084289X.rd448.pub2
- ↑ H. Suzuki; H. Tani (1984). "A mild cyclization of 2-aminoalcohols to aziridines using diphosphorus tetraiodide". Chemistry Letters. 13 (12): 2129–2130. doi:10.1246/cl.1984.2129.
- ↑ Vikas N. Telvekar; Somsundaram N. Chettiar (June 2007). "A novel system for decarboxylative bromination". Tetrahedron Letters. 48 (26): 4529–4532. doi:10.1016/j.tetlet.2007.04.137.
- ↑ Richard Kuhn, Alfred Winterstein (1928). "Über konjugierte Doppelbindungen I. Synthese von Diphenyl-poly-enen". Helvetica Chimica Acta. 11 (1): 87–116. doi:10.1002/hlca.19280110107.