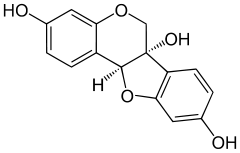
Glycinol (pterocarpan)
For other uses, see Glycinol.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
6H-[1]benzofuro[3,2-c]chromene-3,6a(11aH),9-triol | |
| Identifiers | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 114790 |
| PubChem | 129648 |
| |
| |
| Properties | |
| C15H12O5 | |
| Molar mass | 272.25 g/mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Glycinol is a pterocarpan, a type of natural phenol. It is a phytoalexin found in the soybean (Glycine max). It is formed by the cyclisation of daidzein. It has antiestrogenic activities.[1][2]
It can be synthethised chemically and possesses two chiral centers.[3]
Glycinol is the direct precursor of glyceollins through the action of a prenyltransferase.
Experiments show that the 6a oxygen of glycinol is derived from molecular oxygen.[4]
References
- ↑ Zimmermann, M. C.; Tilghman, S. L.; Boue, S. M.; Salvo, V. A.; Elliott, S.; Williams, K. Y.; Skripnikova, E. V.; Ashe, H.; et al. (2009). "Glyceollin I, a Novel Antiestrogenic Phytoalexin Isolated from Activated Soy". Journal of Pharmacology and Experimental Therapeutics. 332 (1): 35–45. doi:10.1124/jpet.109.160382. PMC 2802480
 . PMID 19797619.
. PMID 19797619. - ↑ Qi Y.; Moco S.I.A.; Boeren S.; Vos C.H. de; Bovy A.G. (2005). "Isolation and identification of glycinol from Glycine max L. Merri". Chinese Journal of Chromatography. 23 (4): 353–357. PMID 16250441.
- ↑ Luniwal Amarjit; Khupse Rahul S; Reese Michael; Lei Fang; Erhardt Paul W (2009). "Total Syntheses of Racemic and Natural Glycinol". Journal of Natural Products. 72 (11): 2072–2075. doi:10.1021/np900509f. PMID 19943626.
- ↑ Matthews, David E.; Plattner, Ronald D.; Vanetten, Hans D. (1989). "The 6a oxygen of the pterocarpan glycinol is derived from molecular oxygen". Phytochemistry. 28: 113. doi:10.1016/0031-9422(89)85020-4.
This article is issued from Wikipedia - version of the 5/31/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.