Heptazine
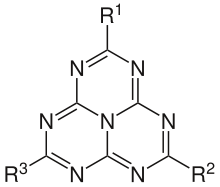
A heptazine, or tri-s-triazine or cyamelurine, is a type of chemical compound that consist of a planar triangular core group, C6N7, or three fused triazine rings, with three substituents at the corners of the triangle.
The general form is 1,3,4,6,7,9,9b-heptaazaphenalene. The parent compound C6N7H3, where the three substituents are hydrogens, is called 1,3,4,6,7,9-hexaazacyc1[3.3.3]azine or tri-s-triazine proper.[1]
Heptazines were discovered in the 19th century but their study has long been hampered by their general insolubility. They are used as flame retardants and have been the object of interest recently for potential applications in electronics materials, explosives, and more.
History
Jöns Jakob Berzelius first mentioned the heptazines in the 1830s when he discovered a polymeric substance after mercury thiocyanate ignition. Justus von Liebig named the polymer melon.[2] Much later in 1937 Linus Pauling showed by x-ray crystallography that heptazines are in fact fused triazines.[2] The unsubstituted heptazine C6N7H3 was synthesized by Ramachandra S. Hosmane and others from the group of N. Leonard in the early 1980s.[1] The structure of Berzelius's melon was confirmed only in 2001.[2][3]
Parent molecule
The parent substance C6N7H3 is a yellow, weakly fluorescent solid with melting point over 300 °C. The compound is generally stable and soluble in organic solvents such as acetonitrile, but is decomposed by water. It has a peculiar crystal structure, whose cell spans 16 molecules in asymmetric positions and orientations.[1][4]
Derivatives and uses
The specific compound with three amino substituents is called melem. When heptazine is polymerized with the tri-s-triazine units linked through an amine (NH) link, it is called melon. Another melamine homologue, but not a heptazine, is the dimer melam, the fused product of 2,4-diamino-6-chloro-s-triazine with melamine. Melem, melon, and melam are effective flame retardant compounds. The compounds have in common that they melt or decompose at very high temperatures and that they are insoluble in any solvent. This makes characterisation difficult.
The heptazine derivate with three hydroxyl substituents is called cyameluric acid and has many possible tautomeric structures. Of the seventeen tautomeric forms, calculations showed that of the tri-oxo forms was found to be the most stable.[5] Therefore, this compound is not a hydroxide but an amide. The first person to suggest a structure for cyameluric acid was J. Loschmidt, as far back as 1861. His structure was in fact a meta-cyclophane, but it is remarkable since at that time cyclic compounds of any type were not widely recognised.[6][7]
The heptazine derivate with an azide substituent and two hydroxyl groups is called Linus Pauling's mystery molecule. It is the last molecule he drew on his chalkboard (preserved for posterity) before he died in 1994. Two theories attempt to lift the mystery. It is suggested that Pauling failed to solve the double helix structure of DNA before Watson and Crick because he viewed uracil as an amide and not as the tautomeric hydroxy compound. The other theory suggests that Pauling intended to use the compound as a potential spectroscopic label for binding to DNA. Nelson Leonard observed that Pauling "must have returned to the source of his original structural inspiration for a new application."[2]
The tri-azido derivatives are investigated for their use as high-energy-density materials (explosives).
It is believed that the graphite form of the carbon nitride C3N4 is built up of linked heptazines. Heptazines could be a precursor molecule to the diamond-like beta carbon nitride.
References
- 1 2 3 Ramachandra S. Hosmane, Mitchell A. Rossman, and Nelson J. Leonard (1982), Synthesis and Structure of Tri-s-triazine J. Am. Chem. Soc., vol. 104 issue 20, pp 5497–5499. doi:10.1021/ja00384a046
- 1 2 3 4 Elizabeth K. Wilson (2004), Old Molecule, New Chemistry. Long-mysterious heptazines are beginning to find use in making carbon nitride materials. Chemical & Engineering News, May 26, 2004. Online version accessed on 2009-06-30.
- ↑ Macromol. Chem. Phys., vol. 202, p. 19 (2001).
- ↑ Manouchehr Shahbaz, Shigeyuki Urano, Pierre R. LeBreton, Mitchell A. Rossman, Ramachandra S. Hosmane, Nelson J. Leonard (1984), Tri-s-triazine: synthesis, chemical behavior, and spectroscopic and theoretical probes of valence orbital structure. J. Am. Chem. Soc., vol. 106 issue 10, pp 2805–2811. doi:10.1021/ja00322a014
- ↑ Ibon Alkorta, Nadine Jagerovic, and José Elguero (2004), Theoretical study of cyameluric acid and related compounds. Arkivoc. Article
- ↑ Henry S. Rzepa, Joseph Loschmidt: Structural formulae, 1861. Accessed on 2009-06-30.
- ↑ Johann Josef Loschmidt (1861), Konstitutionsformeln der Organischen Chemie in Graphischer Darstellung. Nr. 190. Wilhelm Engelmann, Leipzig.