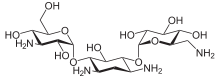
Kanamycin A
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous, intramuscular |
| ATC code | A07AA08 (WHO) J01GB04 (WHO) S01AA24 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | very low after by mouth delivery |
| Metabolism | Unknown |
| Biological half-life | 2 hours 30 minutes |
| Excretion | Urine (as unchanged drug) |
| Identifiers | |
| |
| CAS Number |
59-01-8 |
| PubChem (CID) | 6032 |
| DrugBank |
DB01172 |
| ChemSpider |
5810 |
| UNII |
RUC37XUP2P |
| ChEBI |
CHEBI:17630 |
| ChEMBL |
CHEMBL1384 |
| PDB ligand ID | KAN (PDBe, RCSB PDB) |
| Chemical and physical data | |
| Formula | C18H36N4O11 |
| Molar mass | 484.499 |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Kanamycin, also known as kanamycin A, is an antibiotic used to treat a wide variety of infections. Kanamycin's is a second line treatment of Mycobacterium tuberculosis.[1] It may be used by mouth, injection into a vein, or injection into a muscle. Kanamycin is used for short-term treatment only, usually from 7 to 10 days.[2] It is not effective in treating colds, flu, or viral infections.[2]
Hearing and kidney problems are the most severe adverse effects seen, especially in older people.[3] Kanamycin is not recommended during pregnancy due to potential harm to the baby. Breastfeeding women should also consult with physician before using kanamycin due to its excretion into breast milk and may cause adverse effects in the baby.[3] It works by targeting production of important proteins that are essential for bacterial survival.[3]
It is on the World Health Organization's List of Essential Medicines, the most important medications needed in a basic health system.[4] Kanamycin is isolated from the bacterium Streptomyces kanamyceticus[5] and its most commonly used form is kanamycin sulfate.
Medical uses
Spectrum of activity
Kanamycin is indicated for short term treatment of bacterial infections caused by one or more of the following pathogens: E. coli, Proteus species (both indole-positive and indole-negative), Enterobacter aerogenes, Klebsiella pneumoniae, Serratia marcescens, and Acinetobacter species. In cases of serious infection when the causative organism is unknown, Kanamycin injection in conjunction with a penicillin- or cephalosporin-type drug may be given initially before obtaining results of susceptibility testing.
Kanamycin does not treat viral infections.[6]
Pregnancy and breast feeding
Kanamycin is pregnancy category D in the United States.[6]
Kanamycin is excreted into breast milk in small amounts. The manufacturer recommends that because of the potential for serious adverse effects in nursing infants, the patient should discontinue nursing or discontinue kanamycin, depending on how important the drug is to the mother. The American Academy of Pediatrics considers kanamycin compatible for breast feeding.[7]
Children
Kanamycin should be used with caution in newborns due to the risk of increased serum half-life and drug concentration resulting from immature kidney function.[6]
Side effects
Serious side effects include ringing in the ears or loss of hearing, toxicity to kidneys, and allergic reactions to the drug.[8]
Other side effects include:[6]
Gastrointestinal effects
- Nausea, vomiting, diarrhea
Musculoskeletal effects
- Myasthenia gravis
Neurologic effects
- Headache
- Paresthesias
- Blurring of vision
- Neuromuscular blockade
Metabolic effects
- Malabsorption syndrome
Mechanism
Kanamycin interacts with the 30S subunit of prokaryotic ribosomes. It gives birth to substantial amounts of mistranslation and indirectly inhibits translocation during protein synthesis.[9][10]
Kanamycin works by interfering with protein synthesis. It binds to the 30S subunit of the bacterial ribosome. This results in incorrect alignment with the mRNA and eventually leads to a misread that causes the wrong amino acid to be placed into the peptide. This leads to nonfunctional peptide chains.[11]
Composition
Kanamycin is a mixture of three main components: kanamycin A, B, and C. Kanamycin A is the major component in kanamycin.[12] The effects of these components do not appear to be widely studied as individual compounds when used against prokaryotic and eukaryotic cells.
Biosynthesis
While the main product produced by Streptomyces kanamyceticus is kanamycin A, additional products are also produced, including kanamycin B, kanamycin C, kanamycin D and kanamycin X.
The kanamycin biosynthetic pathway can be divided into two parts. The first part is common to several aminoglycoside antibiotics, such as butirosin and neomycin. In it a unique aminocyclitol, 2-deoxystreptamine, is biosynthesized from D-glucopyranose 6-phosphate in four steps. At this point the kanamycin pathway splits into two branches due to the promiscuity of the next enzyme, which can utilize two different glycosyl donors - UDP-N-acetyl-α-D-glucosamine and UDP-α-D-glucose. One of the branches forms kanamycin C and kanamycin B, while the other branch forms kanamycin D and kanamycin X. However, both kanamycin B and kanamycin D can be converted to kanamycin A, so both branches of the pathway converge at kanamycin A.[13]
Use in research
Kanamycin is used in molecular biology as a selective agent most commonly to isolate bacteria (e.g., E. coli) which have taken up genes (e.g., of plasmids) coupled to a gene coding for kanamycin resistance (primarily Neomycin phosphotransferase II [NPT II/Neo]). Bacteria that have been transformed with a plasmid containing the kanamycin resistance gene are plated on kanamycin (50-100 ug/ml) containing agar plates or are grown in media containing kanamycin (50-100 ug/ml). Only the bacteria that have successfully taken up the kanamycin resistance gene become resistant and will grow under these conditions. As a powder, kanamycin is white to off-white and is soluble in water (50 mg/ml).
At least one such gene, Atwbc19[14] is native to a plant species, of comparatively large size and its coded protein acts in a manner which decreases the possibility of horizontal gene transfer from the plant to bacteria; it may be incapable of giving resistance to bacteria even if gene transfer occurs.
KanMX marker
The selection marker kanMX is a hybrid gene consisting of a bacterial aminoglycoside phosphotransferase (kanr from transposon Tn903) under control of the strong TEF promoter from Ashbya gossypii.[15][16]
Mammalian cells, yeast, and other eukaryotes acquire resistance to geneticin (= G418, an aminoglycoside antibiotic similar to kanamycin) when transformed with a kanMX marker. In yeast, the kanMX marker avoids the requirement of auxotrophic markers. In addition, the kanMX marker renders E. coli resistant to kanamycin. In shuttle vectors the KanMX cassette is used with an additional bacterial promoter. Several versions of the kanMX cassette are in use, e.g. kanMX1-kanMX6. They primarily differ by additional restriction sites and other small changes around the actual open reading frame.[15][17]
References
- ↑ "Kanamycin (Lexi-Drugs)".
- 1 2 "Kanamycin (By injection)".
- 1 2 3 "Drugs.com".
- ↑ "WHO Model List of EssentialMedicines" (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- ↑ Garrod, L.P., et al.: "Antibiotic and Chemotherapy", page 131. Churchill Livingstone, 1981
- 1 2 3 4 "Kanamycin (By injection)".
- ↑ Briggs, Gerald (2011). Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. Lippincott Williams & Wilkins. p. 787.
- ↑ Consumer Drug Information: Kanamycin, 2 April 2008, retrieved 2008-05-04
- ↑ Pestka, S.: "The Use of Inhibitors in Studies on Protein Synthesis", Methods in Enzymology 30, pp.261-282, 1975
- ↑ Misumi, M. & Tanaka, N.: "Mechanism of Inhibition of Translocation by Kanamycin and Viomycin: A Comparative Study with Fusidic Acid, Biochem.Biophys.Res.Commun. 92, pp.647-654, 1980
- ↑ DrugBank, ed. (2016-08-17). "Kanamycin". DrugBank.
- ↑ United States. National Institutes of Health. Kanamycin Compound Summary. PubChem. Web. 21 Aug. 2012.
- ↑ "kanamycin biosynthesis pathway in MetaCyc". MetaCyc.org. Retrieved 30 September 2014.
- ↑ "Horizontal Gene Transfer: Plant vs. Bacterial Genes for Antibiotic Resistance Scenario's—What's the Difference?". Isb.vt.edu. Retrieved 2013-06-24.
- 1 2 Wach, A.; Brachat, A.; Pöhlmann, R.; Philippsen, P. (1994). "New heterologous modules for classical or PCR-based gene disruptions in Saccharomyces cerevisiae". Yeast. 10 (13): 1793–1808. doi:10.1002/yea.320101310. PMID 7747518.
- ↑ Steiner, S.; Philippsen, P. (1994). "Sequence and promoter analysis of the highly expressed TEF gene of the filamentous fungus Ashbya gossypii". Molecular & general genetics : MGG. 242 (3): 263–271. doi:10.1007/BF00280415. PMID 8107673.
- ↑ Wach, A. (1996). "PCR-synthesis of marker cassettes with long flanking homology regions for gene disruptions in S. Cerevisiae". Yeast. 12 (3): 259–265. doi:10.1002/(SICI)1097-0061(19960315)12:3<259::AID-YEA901>3.0.CO;2-C. PMID 8904338.