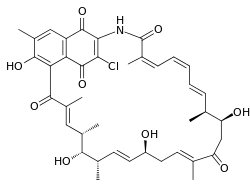
Naphthomycin A
 | |
| Identifiers | |
|---|---|
| 3D model (Jmol) | Interactive image |
| ChemSpider | 8276911 |
| PubChem | 10101379 |
| |
| Properties | |
| C40H46ClNO9 | |
| Molar mass | 720.26 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Naphthomycin A is a type of naphthomycin. It was isolated as a yellow pigment from Streptomyces collinus and it shows antibacterial, antifungal, and antitumor activities.[1] Naphthomycins have the longest ansa aliphatic carbon chain of the ansamycin family.[2] Biosynthetic origins of the carbon skeleton from PKS1 were investigated by feeding 13C-labeled precursors and subsequent 13C-NMR product analysis. Naphthomycin gene clusters have been cloned and sequenced to confirm involvement in biosynthesis via deletion of a 7.2kb region.[3] Thirty-two genes were identified in the 106kb cluster.[4]
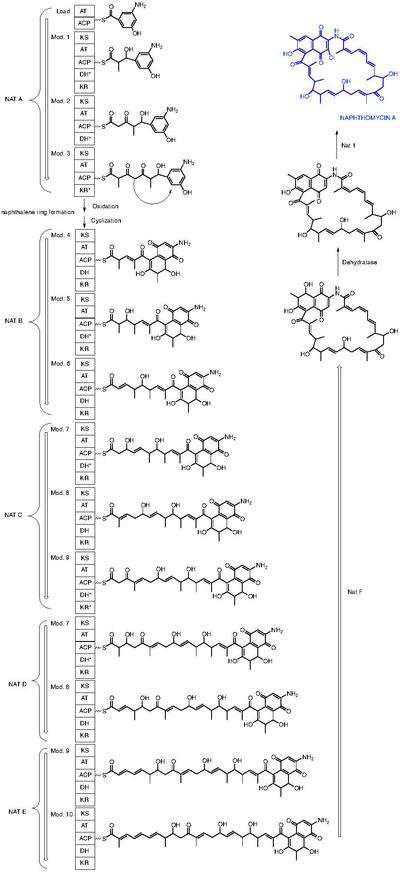
The proposed naphthomycin biosynthetic pathway.[5] A, adenylation; KS, ketosynthase; AT, acyltransferase; T, acyl carrier protein or peptidyl carrier protein; DH, dehydratase; KR, ketoreductase; (*) redundant domain.
References
- ↑ Kang, Q.; Y. Shenb; L. Bai (2012). "Biosynthesis of 3,5-AHBA-derived natural products". Nat. Prod. Rep. 29 (2): 243–263. doi:10.1039/c2np00019a. PMID 22193711.
- ↑ Balerna, M.; W. Keller-Schierlein; C. Martius; H. Zahner (1969). "Stoffwechselprodukte von Mikroorganismen". Arch. Microbiol. 65 (4): 303–317. doi:10.1007/BF00412210. PMID 4988744.
- ↑ Bai, L.; Y. Wu; Q. Kang; Y. Shen; Z. Deng (2012). Patent. CN: 2012-10300294: 225. Missing or empty
|title=(help) - ↑ August, P.R.; L. Tang, Y.J. Yoon, S. Ning, R. MuEller, T.W. Yu, M. Taylor, D. Hoffman, C.G. Kim, X. Zahng, C.R. Hutchinson, H.G. Floss (1998). "Biosynthesis of the ansamycin antibiotic rifamycin: Deductions from the molecular analysis of the rif biosynthetic gene cluster of Amycolatopsis mediterranei S699". Chem. Biol. 5 (2): 69–79. doi:10.1016/S1074-5521(98)90141-7. PMID 9512878. Cite uses deprecated parameter
|coauthors=(help) - ↑ Kang, Q; Y. Shenb; L. Bai (2012). "Biosynthesis of 3,5-AHBA-derived natural products". Nat. Prod. Rep. 29 (2): 243–263. doi:10.1039/c2np00019a. PMID 22193711.
This article is issued from Wikipedia - version of the 12/3/2015. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.