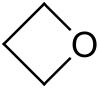
Oxetane
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Oxetane[1] | |||
| Other names
1,3-Propylene oxide 1,3-Epoxypropane Oxacyclobutane Trimethylene oxide | |||
| Identifiers | |||
| 503-30-0 | |||
| 3D model (Jmol) | Interactive image | ||
| ChEBI | CHEBI:30965 | ||
| ChemSpider | 9994 | ||
| ECHA InfoCard | 100.007.241 | ||
| PubChem | 10423 | ||
| UNII | I279Q16FU6 | ||
| |||
| |||
| Properties | |||
| C3H6O | |||
| Molar mass | 58.08 g/mol | ||
| Density | 0.8930 g/cm3 | ||
| Boiling point | 49 to 50 °C (120 to 122 °F; 322 to 323 K) | ||
| Hazards | |||
| Flash point | −28.3 °C; −19.0 °F; 244.8 K (NTP, 1992) | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Oxetane, or 1,3-propylene oxide, is an heterocyclic organic compound with the molecular formula C3H6O, having a four-membered ring with three carbon atoms and one oxygen atom.
The term oxetane may also refer more generally to any organic compound containing an oxetane ring.
Production
A typical well-known method of preparation is the reaction of potassium hydroxide with 3-chloropropyl acetate at 150 °C:[2]
Yield of oxetane made this way is c. 40%, as the synthesis can lead to a variety of by-products.
Other possible reactions to form oxetane ring is the Paternò–Büchi reaction. The oxetane ring can also be formed through diol cyclization as well as through decarboxylation of a six-membered cyclic carbonate.
Taxol
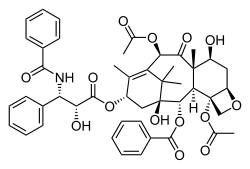
Paclitaxel (Taxol) is an example of a natural product containing an oxetane ring. Taxol has become a major point of interest among researchers due to its unusual structure and success in the involvement of cancer treatment.[3] The attached oxetane ring is an important feature that is used for the binding of microtubules in structure activity; however little is known about how the reaction is catalyzed in nature, which creates a challenge for scientists trying to synthesize the product.[3]
See also
- β-Propiolactone or 2-oxetanone.
- 3-Oxetanone
References
- ↑ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 147. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ↑ C. R. Noller (1955). "Trimethylene Oxide". Org. Synth. 29: 92.; Coll. Vol., 3, p. 835
- 1 2 Willenbring, Dan, and Dean J. Tantillo.. "Mechanistic possibilities for oxetane formation in the biosynthesis of Taxol’s D ring." Russian Journal of General Chemistry 78.4 (Apr. 2008): 7237–31. Advanced Placement Source. EBSCO. [Library name], [City], [State abbreviation]. 22 Apr. 2009 <http://search.ebscohost.com/login.aspx?direct=true&db=aqh&AN=32154883&site=ehost-live>