Phosphite ester
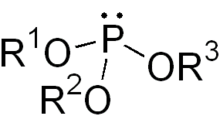
In chemistry a phosphite ester or organophosphite is a type of organophosphorous compound with the general structure P(OR)3. They can be considered as esters of phosphorous acid, H3PO3, with the simplest example being trimethylphosphite, P(OCH3)3.
Synthesis
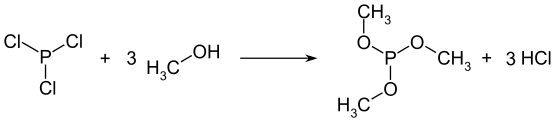
Phosphite esters can be prepared by reacting phosphorus trichloride or phosphorus tribromide with an alcohol. The reaction can be catalysed by the presence of a suitable non-nucleophic base, such as a tertiary amine, with will remove the HCl or HBr by-product.
Structure and bonding
Phosphite esters adopt trigonal pyramidal molecular geometry due to the lone pairs on the phosphorous. As such they typically display C3v molecular symmetry.
Reactions
Phosphite esters are used in the Perkow reaction in the formation of vinyl phosphonates and in the Michaelis–Arbuzov reaction to form phosphonates. One other particular use is that of reducing agent. For example, triethylphosphite is known to reduce certain hydroperoxides to alcohols formed by autoxidation[1] (scheme 1). In this process the phosphite is converted to a phosphate ester.

This reaction type is also utilized in the Wender Taxol total synthesis.
Coordination chemistry
Phosphite esters are lewis basic and hence can form coordination complexes with various metal ions. When coordinated their trigonal pyramidal molecular geometry results in a smaller ligand cone angles than might be expected, making them appealing as ligands. However they remain less important that the structurally related phosphine ligand family.
See also
- Phosphinite P(OR)R2
- Phosphonite P(OR)2R
- Ortho ester CH(OR)3
- Borate ester B(OR)3
- Trimethylolpropane phosphite a phosphite ligand used in organometallic chemistry.
References
- ↑ J. N. Gardner, F. E. Carlon and O. Gnoj (1968). "One-step procedure for the preparation of tertiary α-ketols from the corresponding ketones". J. Org. Chem. 33 (8): 3294–3297. doi:10.1021/jo01272a055.