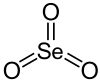
Selenium trioxide
| |||
| Identifiers | |||
|---|---|---|---|
| 13768-86-0 | |||
| 3D model (Jmol) | Interactive image | ||
| ChemSpider | 103019 | ||
| ECHA InfoCard | 100.033.972 | ||
| PubChem | 115128 | ||
| |||
| |||
| Properties | |||
| SeO3 | |||
| Molar mass | 126.96 g/mol | ||
| Appearance | white hygroscopic crystals | ||
| Density | 3.44 g/cm3 | ||
| Melting point | 118.35 °C (245.03 °F; 391.50 K) | ||
| Boiling point | sublimes | ||
| very soluble | |||
| Structure | |||
| tetragonal | |||
| Hazards | |||
| EU classification (DSD) |
not listed | ||
| Lethal dose or concentration (LD, LC): | |||
| LD50 (median dose) |
7 mg/kg (rat, oral) 7.08 mg/kg (mouse, oral) 5.06 mg/kg (guinea pig, oral) 2.25 mg/kg (rabbit, oral) 13 mg/kg (horse, oral)[2] | ||
| LC50 (median concentration) |
13 mg/kg (pig, oral) 9.9 mg/kg (cow, oral) 3.3 mg/kg (goat, oral) 3.3 mg/kg (sheep, oral)[2] | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Selenium trioxide is the inorganic compound with the formula SeO3. It is white, hygroscopic solid. It is also an oxidizing agent and a Lewis acid. It is of academic interest as a precursor to Se(VI) compounds.[3]
Preparation
Selenium trioxide is difficult to prepare because it is unstable with respect to the dioxide:
- 2 SeO3 → 2 SeO2 + O2
It has been generated in a number of ways despite the fact that the dioxide does not combust under normal conditions.[3] One method entails dehydration of anhydrous selenic acid with phosphorus pentoxide at 150-160 °C. The reaction of liquid sulfur trioxide with potassium selenate
- SO3 + K2SeO4 → K2SO4 + SeO3
Reactions
In its chemistry SeO3 generally resembles sulfur trioxide, SO3, rather than tellurium trioxide, TeO3.[3]
At 120 °C SeO3 reacts with selenium dioxide to form the Se(VI)-Se(IV) compound diselenium pentoxide:[4]
- SeO3 + SeO2 → Se2O5
It reacts with selenium tetrafluoride to form selenoyl fluoride, the selenium analogue of sulfuryl fluoride
- SeO3 + SeF4 → SeO2F2
As with SO3 adducts are formed with Lewis bases such as pyridine, dioxane and ether.[3]
With lithium oxide and sodium oxide it reacts to form salts of SeVIO54− and SeVIO66−:[5] With Li2O, it gives Li4SeO5, containing the trigonal pyramidal anion SeVIO54− with equatorial bonds, 170.6-171.9 pm; and longer axial Se-O bonds of 179.5 pm. With Na2O it gives Na4SeO5, containing the square pyramidal SeVIO54−, with Se-O bond lengths ranging from range 1.729 → 1.815 pm, and Na12(SeO4)3(SeO6), containing octahedral SeVIO66−. SeVIO66− is the conjugate base of the unknown orthoselenic acid (Se(OH)6).
Structure
In the solid phase SeO3 consists of cyclic tetramers, with an 8 membered (Se-O)4 ring. Selenium atoms are 4-coordinate, bond lengths being Se-O bridging are 175 pm and 181pm, non-bridging 156 and 154 pm.[5]
SeO3 in the gas phase consists of tetramers and monomeric SeO3 which is trigonal planar with an Se-O bond length of 168.78 pm.[6]
References
- ↑ Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, FL: CRC Press. pp. 4–81. ISBN 0-8493-0594-2.
- 1 2 "Selenium compounds (as Se)". Immediately Dangerous to Life and Health. National Institute for Occupational Safety and Health (NIOSH).
- 1 2 3 4 Egon Wiberg, Arnold Frederick Holleman (2001) Inorganic Chemistry, Elsevier ISBN 0123526515
- ↑ Z. Žák "Crystal structure of diselenium pentoxide Se2O5" Zeitschrift für anorganische und allgemeine Chemie 1980, volume 460, pp. 81–85. doi:10.1002/zaac.19804600108
- 1 2 Handbook of Chalcogen Chemistry: New Perspectives in Sulfur, Selenium and Tellurium, Franceso A. Devillanova, Royal Society of Chemistry, 2007, ISBN 9780854043668
- ↑ Brassington, N. J.; Edwards, H. G. M.; Long, D. A.; Skinner, M. (1978). "The pure rotational Raman spectrum of SeO3". Journal of Raman Spectroscopy. 7 (3): 158–160. doi:10.1002/jrs.1250070310. ISSN 0377-0486.
Further reading
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9.
- Schmidt, Prof. Dr. Max; Dr. P. Bornmann; Dr. Irmgard Wilhelm (1963-10-02). "The Chemistry of Selenium Trioxide". Angewandte Chemie International Edition in English. 2 (11): 691–692. doi:10.1002/anie.196306913.