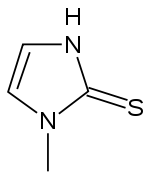
Methimazole
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682464 |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | H03BB02 (WHO) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 93% |
| Protein binding | None |
| Metabolism | Hepatic |
| Biological half-life | 5-6 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number |
60-56-0 |
| PubChem (CID) | 1349907 |
| IUPHAR/BPS | 6649 |
| DrugBank |
DB00763 |
| ChemSpider |
1131173 |
| UNII |
554Z48XN5E |
| KEGG |
D00401 |
| ChEBI |
CHEBI:50673 |
| ChEMBL |
CHEMBL1515 |
| ECHA InfoCard | 100.000.439 |
| Chemical and physical data | |
| Formula | C4H6N2S |
| Molar mass | 114.17 g/mol |
| 3D model (Jmol) | Interactive image |
| Melting point | 146 °C (295 °F) |
| Solubility in water | 275[1] mg/mL (20 °C) |
| |
| |
| (verify) | |
Methimazole (also known as Tapazole or Thiamazole or MMI) is an antithyroid drug,[2] and part of the thioamide group. Like its counterpart propylthiouracil, a major side effect of treatment is agranulocytosis.
Indications
Methimazole is a drug used to treat hyperthyroidism, a condition that occurs when the thyroid gland begins to produce an excess of thyroid hormone. The drug may also be taken before thyroid surgery to lower thyroid hormone levels and minimize the effects of thyroid manipulation. Additionally, methimazole is used in the veterinary setting to treat hyperthyroidism in cats.
Mechanism of action
Methimazole inhibits the enzyme thyroperoxidase, which normally acts in thyroid hormone synthesis by oxidizing the anion iodide (I−) to iodine (I2), hypoiodous acid (HOI), enzyme linked hypoiodate (EOI) facilitating iodine's addition to tyrosine residues on the hormone precursor thyroglobulin, a necessary step in the synthesis of triiodothyronine (T3) and thyroxine (T4).
It does not inhibit the action of the sodium-dependent iodide transporter located on follicular cells' basolateral membranes. Inhibition of this step requires competitive inhibitors such as perchlorate and thiocyanate.
Adverse effects
It is important to monitor any symptoms of fever or sore throat while taking methimazole; this could indicate the development of agranulocytosis, an uncommon but severe side effect resulting from a drop in the white blood cell count (to be specific, neutropenia, a deficiency of neutrophils). A complete blood count (CBC) with differential is performed to confirm the suspicion, in which case the drug is discontinued.[4] Administration of recombinant human granulocyte colony-stimulating factor (rhG-CSF) may increase recovery.
Other known side effects include:
- skin rash
- itching
- abnormal hair loss
- upset stomach
- vomiting
- loss of taste
- abnormal sensations (tingling, prickling, burning, tightness, and pulling)
- swelling
- joint and muscle pain
- drowsiness
- dizziness
- decreased platelet count (thrombocytopenia)
- aplasia cutis congenita (prenatal exposure)
Drug interactions
Adverse effects may occur for individuals who:
- Take anticoagulants ('blood thinners') such as warfarin (Coumadin), diabetes medications, digoxin (Lanoxin), theophylline (Theobid, Theo-Dur), and vitamins
- Have ever had any blood disease, such as decreased white blood cells (leukopenia), decreased platelets (thrombocytopenia) or aplastic anemia, or liver disease (hepatitis, jaundice)
- Are pregnant, or going to become pregnant, or are breast-feeding.
Methimazole has been demonstrated to potently inhibit all CYP450 enzymes, meaning that the plasma concentration of all hepatically metabolized drugs will be significantly increased when taken with, or after, methimazole.[5] This must be considered when starting a patient on methimazole, increasing their dose, or starting a methimazole patient on another drug. Many drugs will need their doses lowered and dosing-time intervals increased when co-administered with methimazole. For some hepatically metabolized drugs with very low therapeutic indexes, co-administration with methimazole may not be possible at all.
See also
References
- ↑ "DrugBank: Methimazole (DB00763)". drugbank.ca. Retrieved 21 July 2015.
- ↑ Nakamura H, Noh JY, Itoh K, Fukata S, Miyauchi A, Hamada N (June 2007). "Comparison of methimazole and propylthiouracil in patients with hyperthyroidism caused by Graves' disease". The Journal of Clinical Endocrinology and Metabolism. 92 (6): 2157–62. doi:10.1210/jc.2006-2135. PMID 17389704.
- ↑ Crescioli C, Cosmi L, Borgogni E, et al. (October 2007). "Methimazole inhibits CXC chemokine ligand 10 secretion in human thyrocytes". J. Endocrinol. 195 (1): 145–55. doi:10.1677/JOE-07-0240. PMID 17911406.
- ↑ Fumarola, A; Di Fiore, A; Dainelli, M; Grani, G; Calvanese, A (Nov 2010). "Medical treatment of hyperthyroidism: state of the art.". Experimental and Clinical Endocrinology & Diabetes. 118 (10): 678–84. doi:10.1055/s-0030-1253420. PMID 20496313.
- ↑ Guo Z, Raeissi S, White RB, Stevens JC (March 1997). "Orphenadrine and methimazole inhibit multiple cytochrome P450 enzymes in human liver microsomes.". Drug Metab. Dispos. 25 (3): 390–3. PMID 9172960.