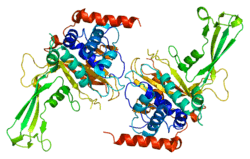
USP7
| View/Edit Human | View/Edit Mouse |
Ubiquitin-specific-processing protease 7 (USP7), also known as ubiquitin carboxyl-terminal hydrolase 7 or herpesvirus-associated ubiquitin-specific protease (HAUSP), is an enzyme that in humans is encoded by the USP7 gene.[3][4][5][6]
Function
Regulation of the p53 tumor suppressor
USP7 or HAUSP is a ubiquitin specific protease or a deubiquitylating enzyme that cleaves ubiquitin from its substrates.[7] Since ubiquitylation (polyubiquitination) is most commonly associated with the stability and degradation of cellular proteins, HAUSP activity generally stabilizes its substrate proteins.
HAUSP is most popularly known as a direct antagonist of Mdm2, the E3 ubiquitin ligase for the tumor suppressor protein, p53.[8] Normally, p53 levels are kept low in part due to Mdm2-mediated ubiquitylation and degradation of p53. In response to oncogenic insults, HAUSP can deubiquitinate p53 and protect p53 from Mdm2-mediated degradation, indicating that it may possess a tumor suppressor function for the immediate stabilization of p53 in response to stress.
Another important role of HAUSP function involves the oncogenic stabilization of p53. Oncogenes such as Myc and E1A are thought to activate p53 through a p19 alternative reading frame (p19ARF, also called ARF)-dependent pathway, although some evidence suggests ARF is not essential in this process. A possibility is that HAUSP provides an alternative pathway for safeguarding the cell against oncogenic insults.
Role in transcriptional regulation
USP7 can deubiquitinate histone H2B and this activity is associated with gene silencing in Drosophila.[9] USP7 associates with a metabolic enzyme, GMP synthetase (GMPS) and this association stimulates USP7 deubiquitinase activity towards H2B.[9] The USP7-GMPS complex is recruited to the polycomb (Pc) region in Drosophila and contributes to epigenetic silecing of homeotic genes.[10]
Association with herpesviruses
USP7 was originally identified as a protein associated with the ICP0 protein of herpes simplex virus (HSV), hence the name Herpesvirus Associated USP (HAUSP). ICP0 is an E3-ubiquitin ligase that is involved in ubiquitination and subsequent degradation of itself and certain cellular proteins. USP7 has been shown to regulate the auto-ubiquitination and degradation of ICP0.
More recently, an interaction between USP7 and the EBNA1 protein of Epstein-Barr virus (EBV) (another herpesvirus) was also discovered.[11] This interaction is particularly interesting given the oncogenic potential (potential to cause cancer) of EBV, which is associated with several human cancers. EBNA1 can compete with p53 for binding USP7. Stabilization by USP7 is important for the tumor suppressor function of p53. In cells, EBNA1 can sequester USP7 from p53 and thus attenuate stabilization of p53, rendering the cells predisposed to turning cancerous. Compromising the function of p53 by sequestering USP7 is one way EBNA1 can contribute to the oncogenic potential of EBV. Additionally, human USP7 was also shown to form a complex with GMPS and this complex is recruited to EBV genome sequences.[12] USP7 was shown to be important for histone H2B deubiquitination in human cells and for deubiquitination of histone H2B incorporated in the EBV genome. Thus USP7 may also be important for regulation of viral gene expression.
The fact that viral proteins have evolved so as to target USP7, underscores the significance of USP7 in tumor suppression and other cellular processes.
Binding partners
The following is a list of some of the known cellular binding partners of USP7/HAUSP:
Interactions
USP7 has been shown to interact with Ataxin 1,[14] CLSPN[15] and P53.[8] A proteomic screen conducted to identify interacting partners of 75 human deubiquitinating enzymes (DUBs) has revealed several novel binding partners of USP7.[16]
References
- ↑ "Human PubMed Reference:".
- ↑ "Mouse PubMed Reference:".
- ↑ Puente XS, Sánchez LM, Overall CM, López-Otín C (Jul 2003). "Human and mouse proteases: a comparative genomic approach". Nature Reviews. Genetics. 4 (7): 544–58. doi:10.1038/nrg1111. PMID 12838346.
- ↑ Robinson PA, Lomonte P, Markham AF, Everett RD (Mar 1999). "Assignment1 of herpesvirus-associated ubiquitin-specific protease gene HAUSP to human chromosome band 16p13.3 by in situ hybridization". Cytogenetics and Cell Genetics. 83 (1-2): 100. doi:10.1159/000015142. PMID 9925944.
- ↑ "Entrez Gene: USP7 ubiquitin specific peptidase 7 (herpes virus-associated)".
- ↑ Everett RD, Meredith M, Orr A, Cross A, Kathoria M, Parkinson J (Apr 1997). "A novel ubiquitin-specific protease is dynamically associated with the PML nuclear domain and binds to a herpesvirus regulatory protein". The EMBO Journal. 16 (7): 1519–30. doi:10.1093/emboj/16.7.1519. PMC 1169756
 . PMID 9130697.
. PMID 9130697. - ↑ Holowaty MN, Sheng Y, Nguyen T, Arrowsmith C, Frappier L (Nov 2003). "Protein interaction domains of the ubiquitin-specific protease, USP7/HAUSP". The Journal of Biological Chemistry. 278 (48): 47753–61. doi:10.1074/jbc.M307200200. PMID 14506283.
- 1 2 Li M, Chen D, Shiloh A, Luo J, Nikolaev AY, Qin J, Gu W (Apr 2002). "Deubiquitination of p53 by HAUSP is an important pathway for p53 stabilization". Nature. 416 (6881): 648–53. doi:10.1038/nature737. PMID 11923872.
- 1 2 van der Knaap JA, Kumar BR, Moshkin YM, Langenberg K, Krijgsveld J, Heck AJ, Karch F, Verrijzer CP (Mar 2005). "GMP synthetase stimulates histone H2B deubiquitylation by the epigenetic silencer USP7". Molecular Cell. 17 (5): 695–707. doi:10.1016/j.molcel.2005.02.013. PMID 15749019.
- ↑ van der Knaap JA, Kozhevnikova E, Langenberg K, Moshkin YM, Verrijzer CP (Feb 2010). "Biosynthetic enzyme GMP synthetase cooperates with ubiquitin-specific protease 7 in transcriptional regulation of ecdysteroid target genes". Molecular and Cellular Biology. 30 (3): 736–44. doi:10.1128/MCB.01121-09. PMC 2812229
 . PMID 19995917.
. PMID 19995917. - ↑ Holowaty MN, Frappier L (Nov 2004). "HAUSP/USP7 as an Epstein-Barr virus target". Biochemical Society Transactions. 32 (Pt 5): 731–2. doi:10.1042/BST0320731. PMID 15494000.
- ↑ Sarkari F, Sanchez-Alcaraz T, Wang S, Holowaty MN, Sheng Y, Frappier L (Oct 2009). Speck SH, ed. "EBNA1-mediated recruitment of a histone H2B deubiquitylating complex to the Epstein-Barr virus latent origin of DNA replication". PLoS Pathogens. 5 (10): e1000624. doi:10.1371/journal.ppat.1000624. PMC 2757719
 . PMID 19834552.
. PMID 19834552. - ↑ Schwertman P, Lagarou A, Dekkers DH, Raams A, van der Hoek AC, Laffeber C, Hoeijmakers JH, Demmers JA, Fousteri M, Vermeulen W, Marteijn JA (May 2012). "UV-sensitive syndrome protein UVSSA recruits USP7 to regulate transcription-coupled repair". Nature Genetics. 44 (5): 598–602. doi:10.1038/ng.2230. PMID 22466611.
- ↑ Hong S, Kim SJ, Ka S, Choi I, Kang S (Jun 2002). "USP7, a ubiquitin-specific protease, interacts with ataxin-1, the SCA1 gene product". Molecular and Cellular Neurosciences. 20 (2): 298–306. doi:10.1006/mcne.2002.1103. PMID 12093161.
- ↑ Faustrup H, Bekker-Jensen S, Bartek J, Lukas J, Mailand N (Jan 2009). "USP7 counteracts SCFbetaTrCP- but not APCCdh1-mediated proteolysis of Claspin". The Journal of Cell Biology. 184 (1): 13–9. doi:10.1083/jcb.200807137. PMC 2615094
 . PMID 19124652.
. PMID 19124652. - ↑ Sowa ME, Bennett EJ, Gygi SP, Harper JW (Jul 2009). "Defining the human deubiquitinating enzyme interaction landscape". Cell. 138 (2): 389–403. doi:10.1016/j.cell.2009.04.042. PMC 2716422
 . PMID 19615732.
. PMID 19615732.
Further reading
- D'Andrea A, Pellman D (1999). "Deubiquitinating enzymes: a new class of biological regulators". Critical Reviews in Biochemistry and Molecular Biology. 33 (5): 337–52. doi:10.1080/10409239891204251. PMID 9827704.
- Holowaty MN, Frappier L (Nov 2004). "HAUSP/USP7 as an Epstein-Barr virus target". Biochemical Society Transactions. 32 (Pt 5): 731–2. doi:10.1042/BST0320731. PMID 15494000.
- Everett RD, Meredith M, Orr A, Cross A, Kathoria M, Parkinson J (Feb 1997). "A novel ubiquitin-specific protease is dynamically associated with the PML nuclear domain and binds to a herpesvirus regulatory protein". The EMBO Journal. 16 (3): 566–77. doi:10.1093/emboj/16.3.566. PMC 1169660
 . PMID 9034339.
. PMID 9034339. - Everett RD, Meredith M, Orr A, Cross A, Kathoria M, Parkinson J (Apr 1997). "A novel ubiquitin-specific protease is dynamically associated with the PML nuclear domain and binds to a herpesvirus regulatory protein". The EMBO Journal. 16 (7): 1519–30. doi:10.1093/emboj/16.7.1519. PMC 1169756
 . PMID 9130697.
. PMID 9130697. - Zapata JM, Pawlowski K, Haas E, Ware CF, Godzik A, Reed JC (Jun 2001). "A diverse family of proteins containing tumor necrosis factor receptor-associated factor domains". The Journal of Biological Chemistry. 276 (26): 24242–52. doi:10.1074/jbc.M100354200. PMID 11279055.
- Li M, Chen D, Shiloh A, Luo J, Nikolaev AY, Qin J, Gu W (Apr 2002). "Deubiquitination of p53 by HAUSP is an important pathway for p53 stabilization". Nature. 416 (6881): 648–53. doi:10.1038/nature737. PMID 11923872.
- Hong S, Kim SJ, Ka S, Choi I, Kang S (Jun 2002). "USP7, a ubiquitin-specific protease, interacts with ataxin-1, the SCA1 gene product". Molecular and Cellular Neurosciences. 20 (2): 298–306. doi:10.1006/mcne.2002.1103. PMID 12093161.
- Hu M, Li P, Li M, Li W, Yao T, Wu JW, Gu W, Cohen RE, Shi Y (Dec 2002). "Crystal structure of a UBP-family deubiquitinating enzyme in isolation and in complex with ubiquitin aldehyde". Cell. 111 (7): 1041–54. doi:10.1016/S0092-8674(02)01199-6. PMID 12507430.
- Holowaty MN, Sheng Y, Nguyen T, Arrowsmith C, Frappier L (Nov 2003). "Protein interaction domains of the ubiquitin-specific protease, USP7/HAUSP". The Journal of Biological Chemistry. 278 (48): 47753–61. doi:10.1074/jbc.M307200200. PMID 14506283.
- Brajenovic M, Joberty G, Küster B, Bouwmeester T, Drewes G (Mar 2004). "Comprehensive proteomic analysis of human Par protein complexes reveals an interconnected protein network". The Journal of Biological Chemistry. 279 (13): 12804–11. doi:10.1074/jbc.M312171200. PMID 14676191.
- Li M, Brooks CL, Kon N, Gu W (Mar 2004). "A dynamic role of HAUSP in the p53-Mdm2 pathway". Molecular Cell. 13 (6): 879–86. doi:10.1016/S1097-2765(04)00157-1. PMID 15053880.
- Cummins JM, Rago C, Kohli M, Kinzler KW, Lengauer C, Vogelstein B (Apr 2004). "Tumour suppression: disruption of HAUSP gene stabilizes p53". Nature. 428 (6982): 1 p following 486. doi:10.1038/nature02501. PMID 15058298.
- Canning M, Boutell C, Parkinson J, Everett RD (Sep 2004). "A RING finger ubiquitin ligase is protected from autocatalyzed ubiquitination and degradation by binding to ubiquitin-specific protease USP7". The Journal of Biological Chemistry. 279 (37): 38160–8. doi:10.1074/jbc.M402885200. PMID 15247261.
- Beausoleil SA, Jedrychowski M, Schwartz D, Elias JE, Villén J, Li J, Cohn MA, Cantley LC, Gygi SP (Aug 2004). "Large-scale characterization of HeLa cell nuclear phosphoproteins". Proceedings of the National Academy of Sciences of the United States of America. 101 (33): 12130–5. doi:10.1073/pnas.0404720101. PMC 514446
 . PMID 15302935.
. PMID 15302935. - Saridakis V, Sheng Y, Sarkari F, Holowaty MN, Shire K, Nguyen T, Zhang RG, Liao J, Lee W, Edwards AM, Arrowsmith CH, Frappier L (Apr 2005). "Structure of the p53 binding domain of HAUSP/USP7 bound to Epstein-Barr nuclear antigen 1 implications for EBV-mediated immortalization". Molecular Cell. 18 (1): 25–36. doi:10.1016/j.molcel.2005.02.029. PMID 15808506.