Woollins' reagent
 | |
 | |
| Names | |
|---|---|
| IUPAC names
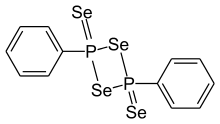
2,4-Diphenyl-1,3,2,4- diselenadiphosphetan-2,4-diselenide | |
| Other names
Woollins' Reagent | |
| Identifiers | |
| 122039-27-4 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 4242993 |
| ECHA InfoCard | 100.155.582 |
| PubChem | 5066075 |
| |
| |
| Properties | |
| C12H10P2Se4 | |
| Molar mass | 532.04 g·mol−1 |
| Appearance | red powder |
| Melting point | 192 to 204 °C (378 to 399 °F; 465 to 477 K) [1] |
| soluble in toluene at elevated temperatures | |
| Hazards | |
| R-phrases | R23/25 R33 R50/53 |
| S-phrases | (S1/2) S20/21 S28 S45 S60 S61 |
| Related compounds | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Woollins' reagent is an organic compound containing phosphorus and selenium. Analogous to Lawesson's reagent, it is used mainly as a selenation reagent. It is named after Professor John Derek Woollins, who is currently the Vice Principal of Research at the University of St Andrews.[2]
Preparation
Woollins' reagent is commercially available. It can also be conveniently prepared in the laboratory by heating a mixture of dichlorophenylphosphine and sodium selenide (Na2Se), (itself prepared from reacting elementary selenium with sodium in liquid ammonia).[3] An alternative synthesis is the reaction of the pentamer (PPh)5 with elemental selenium.[4]
Applications
The main use of Woollins' reagent is the selenation of carbonyl compounds.[5] For instance, Woollins' reagent will convert a carbonyl into a selenocarbonyl. Additionally, Woollins' reagent has been used to selenonate carboxylic acids, alkenes, alkynes, and nitriles.[6]
References
- ↑ The Merck Index. An Encyclopaedia of Chemicals, Drugs and Biologicals. 14. Auflage, 2006, S. 1731, ISBN 978-0-911910-00-1.
- ↑ http://chemistry.st-andrews.ac.uk/staff/jdw/group/home.html
- ↑ Ian P. Gray, Pravat Bhattacharyya, Alexandra M. Z. Slawin and J. Derek Woollins (2005). "A New Synthesis of (PhPSe2)2 (Woollins Reagent) and Its Use in the Synthesis of Novel P-Se Heterocycles". Chem. Eur. J. 11 (21): 6221–7. doi:10.1002/chem.200500291. PMID 16075451.
- ↑ M. J. Pilkington, Alexandra M. Z. Slawin and J. Derek Woollins (1990). "The preparation and characterization of binary phosphorus-selenium rings". Heteroatom Chemistry. 1 (5): 351. doi:10.1002/hc.520010502.
- ↑ Pravat Bhattacharyya & J. Derek Woollins (2001). "Selenocarbonyl synthesis using Woollins reagent". Tet. Lett. 42 (34): 5949. doi:10.1016/S0040-4039(01)01113-3.
- ↑ Guoxiong Hua & J. Derek Woollins (2009). "Formation and Reactivity of Phosphorus-Selenium Rings". Angew. Chem. Int. Ed. 48 (8): 1368–1377. doi:10.1002/anie.200800572. PMID 19053094.