5-MeO-MPMI
 | |
| Identifiers | |
|---|---|
| |
| CAS Number |
143321-57-7 |
| PubChem (CID) | 9881324 |
| ChemSpider |
8057000 |
| ChEMBL |
CHEMBL137485 |
| Chemical and physical data | |
| Formula | C15H20N2O |
| Molar mass | 244.332 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
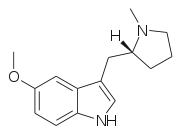
5-MeO-MPMI (also known as 5-Methoxy-N-methyl-(α,N-trimethylene)tryptamine) is a tryptamine derivative that is a psychedelic drug. It was first developed by the team led by JE Macor in 1992,[1] and subsequently investigated by the team led by David Nichols from Purdue University in the late 1990s. This compound produces psychedelic-appropriate responding in animal tests with a similar potency to the amphetamine-derived psychedelic DOI, and has two enantiomers, with only the (R)-enantiomer being active.[2]
See also
References
- ↑ Macor JE, Blake J, Fox CB, Johnson C, Koe BK, Lebel LA, Morrone JM, Ryan K, Schmidt AW, Schulz DW, et al. (1992). "Synthesis and serotonergic pharmacology of the enantiomers of 3-[(N-methylpyrrolidin-2-yl)methyl]-5-methoxy-1H-indole: discovery of stereogenic differentiation in the aminoethyl side chain of the neurotransmitter serotonin". Journal of Medicinal Chemistry. 35 (23): 4503–4305. doi:10.1021/jm00101a032. PMID 1447752.
- ↑ Gerasimov M, Marona-Lewicka D, Kurrasch-Orbaugh DM, Qandil AM, Nichols DE (1999). "Further studies on oxygenated tryptamines with LSD-like activity incorporating a chiral pyrrolidine moiety into the side chain". Journal of Medicinal Chemistry. 42 (20): 4257–4263. doi:10.1021/jm990325u. PMID 10514296.
This article is issued from Wikipedia - version of the 7/4/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.