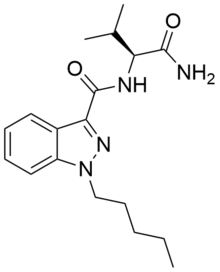
AB-PINACA
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
1445752-09-9 |
| PubChem (CID) | 71301472 |
| ChemSpider | 28537615 |
| Chemical and physical data | |
| Formula | C18H26N4O2 |
| Molar mass | 330.43 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
AB-PINACA is a compound that was first identified as a component of synthetic cannabis products in Japan in 2012.[1]
It was originally developed by Pfizer in 2009 as an analgesic medication.[2][3]
AB-PINACA acts as a potent agonist for the CB1 receptor (Ki = 2.87 nM, EC50 = 1.2 nM) and CB2 receptor (Ki = 0.88 nM, EC50 = 2.5 nM) and fully substitutes for Δ9-THC in rat discrimination studies, while being 1.5x more potent.[4][5]
Side effects
There have been a number of reported cases of deaths and hospitalizations in relation to this synthetic cannabinoid.[6]
Legality
AB-PINACA is an Anlage II controlled substance in Germany as of November 2014.[7]
AB-PINACA is listed in the Fifth Schedule of the Misuse of Drugs Act (MDA) and therefore illegal in Singapore as of May 2015.[8]
It's a Schedule I controlled substance in the USA.[9]
As of October 2015 AB-PINACA is a controlled substance in China.[10]
See also
References
- ↑ Uchiyama, N.; Matsuda, S.; Wakana, D.; Kikura-Hanajiri, R.; Goda, Y. (2012). "New cannabimimetic indazole derivatives, N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-pentyl-1H-indazole-3-carboxamide (AB-PINACA) and N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-(4-fluorobenzyl)-1H-indazole-3-carboxamide (AB-FUBINACA) identified as designer drugs in illegal products". Forensic Toxicology. 31: 93–100. doi:10.1007/s11419-012-0171-4.
- ↑ "AB-PINACA". Cayman Chemical. Retrieved 25 June 2015.
- ↑ "Patent WO/2009/106980 - Indazole derivatives".
- ↑ Banister, Samuel D.; Moir, Michael; Stuart, Jordyn; Kevin, Richard C.; Wood, Katie E.; Longworth, Mitchell; Wilkinson, Shane M.; Beinat, Corinne; Buchanan, Alexandra S.; Glass, Michelle; Connor, Mark; McGregor, Iain S.; Kassiou, Michael (2015). "Pharmacology of Indole and Indazole Synthetic Cannabinoid Designer Drugs AB-FUBINACA, ADB-FUBINACA, AB-PINACA, ADB-PINACA, 5F-AB-PINACA, 5F-ADB-PINACA, ADBICA, and 5F-ADBICA". ACS Chemical Neuroscience. 6 (9): 1546–59. doi:10.1021/acschemneuro.5b00112. PMID 26134475.
- ↑ Jenny L Wiley; Julie A Marusich; Timothy W Lefever; Kateland R Antonazzo; Michael T Wallgren; Ricardo A Cortes; Purvi R Patel; Megan Grabenauer; Katherine N Moore; Brian F Thomas (June 2015). "AB-CHMINACA, AB-PINACA, and FUBIMINA: Affinity and Potency of Novel Synthetic Cannabinoids in Producing Δ9-Tetrahydrocannabinol-Like Effects in Mice". Journal of Pharmacology and Experimental Therapeutics. 354 (3): 328–39. doi:10.1124/jpet.115.225326. PMC 4538877
 . PMID 26105953.
. PMID 26105953. - ↑ Jordan Trecki; Roy R. Gerona; Michael D. Schwartz (July 2015). "Synthetic Cannabinoid–Related Illnesses and Deaths". New England Journal of Medicine. 373 (2): 103–107. doi:10.1056/NEJMp1505328. PMID 26154784.
- ↑ "Gesetz über den Verkehr mit Betäubungsmitteln (Betäubungsmittelgesetz - BtMG) Anlage II (zu § 1 Abs. 1) (verkehrsfähige, aber nicht verschreibungsfähige Betäubungsmittel)". Retrieved 22 June 2015.
- ↑ "CNB NEWS RELEASE". Central Narcotics Bureau (CNB). 30 April 2015. Retrieved 24 July 2015.
- ↑ "Schedules of controlled substances: Temporary placement of three synthetic cannabinoids into schedule I. Final order" (PDF). Drug Enforcement Administration, Department of Justice. 30 January 2015. Retrieved 9 July 2015.
- ↑ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. 27 September 2015. Retrieved 1 October 2015.