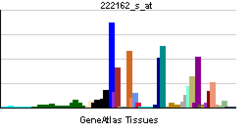
ADAMTS1
| View/Edit Human | View/Edit Mouse |
A disintegrin and metalloproteinase with thrombospondin motifs 1 is an enzyme that in humans is encoded by the ADAMTS1 gene.[3][4]
Function
This gene encodes a member of the ADAMTS (a disintegrin and metalloproteinase with thrombospondin motif) protein family. Members of the family share several distinct protein modules, including a propeptide region, a metalloproteinase domain, a disintegrin-like domain, and a thrombospondin type 1 (TS) motif. Individual members of this family differ in the number of C-terminal TS motifs, and some have unique C-terminal domains. The protein encoded by this gene contains two disintegrin loops and three C-terminal TS motifs and has anti-angiogenic activity. The expression of this gene may be associated with various inflammatory processes as well as development of cancer cachexia. This gene is likely to be necessary for normal growth, fertility, and organ morphology and function.[4]
Interactions
ADAMTS1 has been shown to interact with Vascular endothelial growth factor A.[5]
References
- ↑ "Human PubMed Reference:".
- ↑ "Mouse PubMed Reference:".
- ↑ Vazquez F, Hastings G, Ortega MA, Lane TF, Oikemus S, Lombardo M, Iruela-Arispe ML (September 1999). "METH-1, a human ortholog of ADAMTS-1, and METH-2 are members of a new family of proteins with angio-inhibitory activity". J Biol Chem. 274 (33): 23349–57. doi:10.1074/jbc.274.33.23349. PMID 10438512.
- 1 2 "Entrez Gene: ADAMTS1 ADAM metallopeptidase with thrombospondin type 1 motif, 1".
- ↑ Luque A, Carpizo DR, Iruela-Arispe ML (June 2003). "ADAMTS1/METH1 inhibits endothelial cell proliferation by direct binding and sequestration of VEGF165". J. Biol. Chem. 278 (26): 23656–65. doi:10.1074/jbc.M212964200. PMID 12716911.
Further reading
- Tang BL, Hong W (1999). "ADAMTS: a novel family of proteases with an ADAM protease domain and thrombospondin 1 repeats". FEBS Lett. 445 (2–3): 223–5. doi:10.1016/S0014-5793(99)00119-2. PMID 10094461.
- Hirohata S (2002). "[ADAMTS family--new extracellular matrix degrading enzyme]". Seikagaku. 73 (11): 1333–7. PMID 11831030.
- Kuno K, Kanada N, Nakashima E, et al. (1997). "Molecular cloning of a gene encoding a new type of metalloproteinase-disintegrin family protein with thrombospondin motifs as an inflammation associated gene". J. Biol. Chem. 272 (1): 556–62. doi:10.1074/jbc.272.1.556. PMID 8995297.
- Kuno K, Matsushima K (1998). "ADAMTS-1 protein anchors at the extracellular matrix through the thrombospondin type I motifs and its spacing region". J. Biol. Chem. 273 (22): 13912–7. doi:10.1074/jbc.273.22.13912. PMID 9593739.
- Kuno K, Terashima Y, Matsushima K (1999). "ADAMTS-1 is an active metalloproteinase associated with the extracellular matrix". J. Biol. Chem. 274 (26): 18821–6. doi:10.1074/jbc.274.26.18821. PMID 10373500.
- Nagase T, Kikuno R, Ishikawa KI, et al. (2000). "Prediction of the coding sequences of unidentified human genes. XVI. The complete sequences of 150 new cDNA clones from brain which code for large proteins in vitro". DNA Res. 7 (1): 65–73. doi:10.1093/dnares/7.1.65. PMID 10718198.
- Glienke J, Schmitt AO, Pilarsky C, et al. (2000). "Differential gene expression by endothelial cells in distinct angiogenic states". Eur. J. Biochem. 267 (9): 2820–30. doi:10.1046/j.1432-1327.2000.01325.x. PMID 10785405.
- Shindo T, Kurihara H, Kuno K, et al. (2000). "ADAMTS-1: a metalloproteinase-disintegrin essential for normal growth, fertility, and organ morphology and function". J. Clin. Invest. 105 (10): 1345–52. doi:10.1172/JCI8635. PMC 315464
 . PMID 10811842.
. PMID 10811842. - Hattori M, Fujiyama A, Taylor TD, et al. (2000). "The DNA sequence of human chromosome 21". Nature. 405 (6784): 311–9. doi:10.1038/35012518. PMID 10830953.
- Kuno K, Okada Y, Kawashima H, et al. (2000). "ADAMTS-1 cleaves a cartilage proteoglycan, aggrecan". FEBS Lett. 478 (3): 241–5. doi:10.1016/S0014-5793(00)01854-8. PMID 10930576.
- Sandy JD, Westling J, Kenagy RD, et al. (2001). "Versican V1 proteolysis in human aorta in vivo occurs at the Glu441-Ala442 bond, a site that is cleaved by recombinant ADAMTS-1 and ADAMTS-4". J. Biol. Chem. 276 (16): 13372–8. doi:10.1074/jbc.M009737200. PMID 11278559.
- Wei P, Zhao YG, Zhuang L, et al. (2002). "Protein engineering and properties of human metalloproteinase and thrombospondin 1". Biochem. Biophys. Res. Commun. 293 (1): 478–88. doi:10.1016/S0006-291X(02)00255-3. PMID 12054626.
- Rodríguez-Manzaneque JC, Westling J, Thai SN, et al. (2002). "ADAMTS1 cleaves aggrecan at multiple sites and is differentially inhibited by metalloproteinase inhibitors". Biochem. Biophys. Res. Commun. 293 (1): 501–8. doi:10.1016/S0006-291X(02)00254-1. PMID 12054629.
- Strausberg RL, Feingold EA, Grouse LH, et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. doi:10.1073/pnas.242603899. PMC 139241
 . PMID 12477932.
. PMID 12477932. - Luque A, Carpizo DR, Iruela-Arispe ML (2003). "ADAMTS1/METH1 inhibits endothelial cell proliferation by direct binding and sequestration of VEGF165". J. Biol. Chem. 278 (26): 23656–65. doi:10.1074/jbc.M212964200. PMID 12716911.
- Russell DL, Doyle KM, Ochsner SA, et al. (2004). "Processing and localization of ADAMTS-1 and proteolytic cleavage of versican during cumulus matrix expansion and ovulation". J. Biol. Chem. 278 (43): 42330–9. doi:10.1074/jbc.M300519200. PMID 12907688.
- Ota T, Suzuki Y, Nishikawa T, et al. (2004). "Complete sequencing and characterization of 21,243 full-length human cDNAs". Nat. Genet. 36 (1): 40–5. doi:10.1038/ng1285. PMID 14702039.
External links
- The MEROPS online database for peptidases and their inhibitors: M12.222
- ADAMTS1 on the Atlas of Genetics and Oncology
- Human ADAMTS1 genome location and ADAMTS1 gene details page in the UCSC Genome Browser.