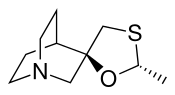
Cevimeline
Cevimeline (trade name Evoxac) is a parasympathomimetic and muscarinic agonist,[1] with particular effect on M1 and M3 receptors. It is used in the treatment of dry mouth and especially associated with Sjögren's syndrome.
Mechanism of action
By activating the M3 receptors of the parasympathetic nervous system, cevimeline stimulates secretion by the salivary glands, thereby alleviating dry mouth.
Side effects
Known side effects include nausea, vomiting, diarrhea, excessive sweating, rash, headache, runny nose, cough, drowsiness, hot flashes, blurred vision, and difficulty sleeping.[2]
Contraindications include asthma and angle closure glaucoma.
See also
- Pilocarpine — a similar parasympathomimetic medication for dry mouth (xerostomia)
- Bethanechol — a similar muscarinic parasympathomimetic with longer-lasting effect
References
External links
|
|---|
|
| |
|---|
| | mACh |
- Muscarinic antagonists: 3-Quinuclidinyl benzilate
- 4-DAMP
- Aclidinium bromide
- Anisodamine
- Anisodine
- Antihistamines (first-generation) (e.g., brompheniramine, chlorphenamine, cyproheptadine, dimenhydrinate, diphenhydramine, doxylamine, mepyramine (pyrilamine), phenindamine, pheniramine, promethazine, tripelennamine, triprolidine)
- Atropine
- Atropine methonitrate
- Atypical antipsychotics (e.g., clozapine, olanzapine, quetiapine, zotepine)
- Benactyzine
- Benzatropine (benztropine)
- Benzilylcholine mustard
- Benzydamine
- BIBN 99
- Biperiden
- Bornaprine
- CAR-226,086
- CAR-301,060
- CAR-302,196
- CAR-302,282
- CAR-302,368
- CAR-302,537
- CAR-302,668
- Caramiphen
- Cloperastine
- CS-27349
- Cyclobenzaprine
- Cyclopentolate
- Darifenacin
- DAU-5884
- Dimethindene
- Dexetimide
- DIBD
- Dicyclomine (dicycloverine)
- Ditran
- EA-3167
- EA-3443
- EA-3580
- EA-3834
- Etanautine
- Etybenzatropine (ethybenztropine)
- Flavoxate
- Himbacine
- HL-031,120
- Ipratropium bromide
- J-104,129
- Hyoscyamine
- Mamba toxin 3
- Mamba toxin 7
- Mazaticol
- Mebeverine
- Methoctramine
- Metixene
- N-Ethyl-3-piperidyl benzilate
- N-Methyl-3-piperidyl benzilate
- Orphenadrine
- Otenzepad
- Oxybutynin
- PBID
- PD-102,807
- PD-0298029
- Phenglutarimide
- Phenyltoloxamine
- Pipenzolate bromide
- Pirenzepine
- Piroheptine
- Procyclidine
- Profenamine
- Revefenacin
- RU-47,213
- SCH-57,790
- SCH-72,788
- SCH-217,443
- Scopolamine (hyoscine)
- Sofpironium bromide
- Solifenacin
- Telenzepine
- Tetracyclic antidepressants (e.g., amoxapine, maprotiline, mianserin, mirtazapine)
- Timepidium bromide
- Tiotropium bromide
- Tolterodine
- Tricyclic antidepressants (e.g., amitriptyline, butriptyline, clomipramine, desipramine, dosulepin (dothiepin), doxepin, imipramine, lofepramine, nortriptyline, protriptyline, trimipramine)
- Trihexyphenidyl
- Tripitamine
- Tropacine
- Tropatepine
- Tropicamide
- Typical antipsychotics (e.g., chlorpromazine, loxapine, thioridazine)
- WIN-2299
- Xanomeline
- Zamifenacin
|
|---|
| | nACh |
- Nicotinic agonists: 5-HIAA
- A-84,543
- A-366,833
- A-582,941
- A-867,744
- ABT-202
- ABT-418
- ABT-560
- ABT-894
- Acetylcholine
- Altinicline
- Anabasine
- Anatoxin-a
- AR-R17779
- Butinoline
- Butyrylcholine
- Carbachol
- Choline
- Cotinine
- Cytisine
- Decamethonium
- Desformylflustrabromine
- Dianicline
- Dimethylphenylpiperazinium
- Epibatidine
- Epiboxidine
- Ethanol
- Ethoxysebacylcholine
- EVP-4473
- EVP-6124
- Galantamine
- GTS-21
- Ispronicline
- Ivermectin
- Levamisole
- Lobeline
- MEM-63,908 (RG-3487)
- Morantel
- Nicotine (tobacco)
- NS-1738
- PHA-543,613
- PHA-709,829
- PNU-120,596
- PNU-282,987
- Pozanicline
- Rivanicline
- RJR-2429
- Sazetidine A
- SB-206553
- Sebacylcholine
- SIB-1508Y
- SIB-1553A
- SSR-180,711
- Suberyldicholine
- Suxamethonium (succinylcholine)
- TC-1698
- TC-1734
- TC-1827
- TC-2216
- TC-5214
- TC-5619
- TC-6683
- Tebanicline
- Tropisetron
- UB-165
- Varenicline
- WAY-317,538
- XY-4083
|
|---|
|
|
|
|
|
| |
|---|
| | ChAT |
- Inhibitors: 1-(-Benzoylethyl)pyridinium
- 2-(α-Naphthoyl)ethyltrimethylammonium
- 3-Chloro-4-stillbazole
- 4-(1-Naphthylvinyl)pyridine
- Acetylseco hemicholinium-3
- Acryloylcholine
- AF64A
- B115
- BETA
- CM-54,903
- N,N-Dimethylaminoethylacrylate
- N,N-Dimethylaminoethylchloroacetate
|
|---|
| | AChE | |
|---|
| | BChE |
- Inhibitors: Cymserine
- Many of the AChE inhibitors listed above
|
|---|
|
|
|
|
|
|