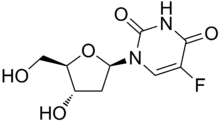
Floxuridine
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682006 |
| Pregnancy category |
|
| Routes of administration | Intra-arterial |
| ATC code | none |
| Identifiers | |
| |
| CAS Number |
50-91-9 |
| PubChem (CID) | 5790 |
| IUPHAR/BPS | 4801 |
| DrugBank |
DB00322 |
| ChemSpider |
5586 |
| UNII |
039LU44I5M |
| KEGG |
D04197 |
| ChEBI |
CHEBI:60761 |
| ChEMBL |
CHEMBL917 |
| ECHA InfoCard | 100.000.066 |
| Chemical and physical data | |
| Formula | C9H11FN2O5 |
| Molar mass | 246.192 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Floxuridine (also 5-fluorodeoxyuridine) is an oncology drug that belongs to the class known as antimetabolites. The drug is most often used in the treatment of colorectal cancer.
Pharmacology
Floxuridine, an analog of 5-fluorouracil, is a fluorinated pyrimidine.
Mechanism of action
Floxuridine works because it is broken down by the body into its active form, which is the same as a metabolite of 5-Fluorouracil.
History
Floxuridine first gained FDA approval in December 1970 under the brand name FUDR. The drug was initially marketed by Roche, which also did a lot of the initial work on 5-fluorouracil. The National Cancer Institute was an early developer of the drug. Roche sold its FUDR product line in 2001 to F H Faulding, which became Mayne Pharma.
Suppliers
In the US the drug is supplied by APP Pharmaceuticals, LLC; Hospira, Inc.; and Bedford Laboratories.