IIG meteorite
| IIG meteorites | |
|---|---|
| — Group — | |
| Type | Iron |
| Structural classification | Hexahedrite |
| Class | Magmatic |
| Subgroups |
|
| Parent body | IIG-IIAB |
| Composition | Meteoric iron (kamacite), nickel (4.1 to 4.9 %), much schreibersite (phosphorus), little sulfur |
| Total known specimens | 6 |
IIG meteorites are a group of iron meteorites. The group currently has six members. They are hexahedrites with large amounts of schreibersite. The meteoric iron is composed of kamacite.[1]
Naming and history
Iron meteorites are designated with a Roman numeral and one or two letters. Classification is based on diagrams in which nickel content of meteoric iron is plotted against trace elements. Clusters in these diagrams are assigned a row (Roman numeral) and a letter in alphabetical order. IIG meteorites are therefore from the second row, cluster G.[2]
The Bellsbank, La Primitiva and Tombigbee meteorites were iron meteorites that were found to have chemical and structural similarities in 1967.[3] Further descriptions were made in 1973 and in 1974 it was proposed that the three meteorites should be grouped into the "Bellsbank Trio" grouplet.[4][5] The group status, that requires five specimen was filled in 1984 by the Twannberg meteorite and in 2000 by the Guanaco meteorite.[6]
Description
IIG meteorites are hexahedrites. The meteoric iron has a low concentration of Nickel (4.1 to 4.9%) and is exclusively kamacite. IIGs contain large amounts of phosphorus in the form of schreibersite and very low concentrations of sulfur.[1][6]
Parent body
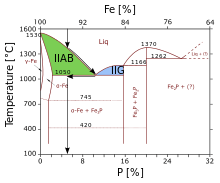
Trace elements of IIAB and IIG meteorites are offset, which was interpreted as the two groups forming on a separate planetesimal. Other explanations for the offset are melt inmiscibility. This process took place while the planetesimal was cooling off. First meteoric iron crystallized into a network of cavities and channels. Eventually crystallization cut off the channels and made cavities of trapped melt. When the remaining melt reached the eutectic point, the cavities crystallized a mixture of schreibersite and meteoric iron. The matrix of this process would form the IIAB meteorites, while the cavities would form the IIG meteorites.[1]
Specimen
The IIG group currently has 6 meteorites that are assigned to it. The Bellsbank, La Primitiva, Tombigbee, Twannberg, Guanaco and the Auburn meteorite.[7]
See also
References
- 1 2 3 4 Wasson, John T.; Choe, Won-Hie (31 July 2009). "The IIG iron meteorites: Probable formation in the IIAB core". Geochimica et Cosmochimica Acta. 73 (16): 4879–4890. doi:10.1016/j.gca.2009.05.062.
- ↑ McSween, Harry Y. (1999). Meteorites and their parent planets (Sec. ed.). Cambridge: Cambridge Univ. Press. ISBN 978-0521587518.
- ↑ Wasson, John T.; Kimbeblin, Jerome (1 October 1967). "The chemical classification of iron meteorites—II. Irons and pallasites with germanium concentrations between 8 and 100 ppm". Geochimica et Cosmochimica Acta. 31 (10): 2065–2093. doi:10.1016/0016-7037(67)90143-3.
- ↑ Scott, Edward R.D; Wasson, John T; Buchwald, V.F (31 July 1973). "The chemical classification of iron meteorites—VII. A reinvestigation of irons with Ge concentrations between 25 and 80 ppm". Geochimica et Cosmochimica Acta. 37 (8): 1957–1983. doi:10.1016/0016-7037(73)90151-8.
- ↑ Wasson, John T. (1974). Meteorites : classification and properties. Berlin: Springer-Verlag. ISBN 978-0387067445.
- 1 2 Hofmann, Beda A.; Lorenzetti, Silvio; Eugster, Otto; Krähenbühl, Urs; Herzog, Gregory; Serefiddin, Feride; Gnos, Edwin; Eggimann, Manuel; Wasson, John T. (1 February 2009). "The Twannberg (Switzerland) IIG iron meteorites: Mineralogy, chemistry, and CRE ages". Meteoritics & Planetary Science. 44 (2): 187–199. doi:10.1111/j.1945-5100.2009.tb00727.x.
- ↑ "Meteoritical Bulletin Database". Meteoritical Society. Retrieved 17 December 2012.