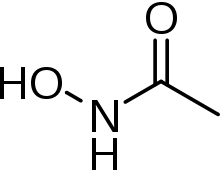
Acetohydroxamic acid
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Consumer Drug Information |
| ATC code | G04BX03 (WHO) |
| Identifiers | |
| |
| CAS Number |
546-88-3 |
| PubChem (CID) | 1990 |
| DrugBank |
DB00551 |
| ChemSpider |
1913 |
| UNII |
4RZ82L2GY5 |
| KEGG |
D00220 |
| ChEBI |
CHEBI:49029 |
| ChEMBL |
CHEMBL734 |
| Chemical and physical data | |
| Formula | C2H5NO2 |
| Molar mass | 75.0666 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Acetohydroxamic acid (also known as AHA or Lithostat) is a drug that is a potent and irreversible inhibitor of bacterial and plant urease usually used for urinary tract infections. The molecule is similar to urea but is not hydrolyzable by the urease enzyme.[1]
Orphan drug
In 1983 the US Food and Drug Administration approved acetohydroxamic acid (AHA) as an orphan drug for "prevention of so-called struvite stones" under the newly enacted Orphan Drug Act of 1983.[2] AHA cannot be patented because it is a standard chemical compound.[2]
References
- ↑ W. Fishbein; P. Carbone (June 1965). "Urease catalysis. ii. Inhibition of the enzyme by hydroxyurea, hydroxylamine, and acetohydroxamic acid". J Biol Chem. 240: 2407–14.
- 1 2 Marwick, Charles (1983). "New drugs selectively inhibit kidney stone formation". The Journal of the American Medical Association. 240 (3): 321–322. doi:10.1001/jama.1983.03340030003001.
See also
This article is issued from Wikipedia - version of the 9/20/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.