Carteolol
 | |
| Clinical data | |
|---|---|
| Trade names | Ocupress |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a601078 |
| Pregnancy category |
|
| Routes of administration | Eye drops |
| ATC code | C07AA15 (WHO) S01ED05 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 85% |
| Metabolism | Hepatic, active with 8-hydrocarteolol |
| Biological half-life | 6–8 hours |
| Excretion | Renal (50-70%) |
| Identifiers | |
| |
| CAS Number | 51781-06-7 |
| PubChem (CID) | 2583 |
| IUPHAR/BPS | 7142 |
| DrugBank |
DB00521 |
| ChemSpider |
2485 |
| UNII |
8NF31401XG |
| KEGG |
D07624 |
| ChEBI |
CHEBI:3437 |
| ChEMBL |
CHEMBL839 |
| Chemical and physical data | |
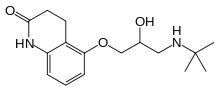
| Formula | C16H24N2O3 |
| Molar mass | 292.373 g/mol |
| 3D model (Jmol) | Interactive image |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Carteolol (trade names Cartrol, Ocupress, Teoptic, Arteolol, Arteoptic, Calte, Cartéabak, Carteol, Cartéol, Cartrol, Elebloc, Endak, Glauteolol, Mikelan, Poenglaucol, Singlauc) is a non-selective beta blocker used to treat glaucoma.
References
External links
- El-Kamel A, Al-Dosari H, Al-Jenoobi F (2006). "Environmentally responsive ophthalmic gel formulation of carteolol hydrochloride.". Drug Deliv. 13 (1): 55–9. doi:10.1080/10717540500309073. PMID 16401594.
- Kuwahara K, Oizumi N, Fujisawa S, Tanito M, Ohira A (2005). "Carteolol hydrochloride protects human corneal epithelial cells from UVB-induced damage in vitro.". Cornea. 24 (2): 213–20. doi:10.1097/01.ico.0000141232.41343.9d. PMID 15725891.
- Trinquand C, Romanet J, Nordmann J, Allaire C (2003). "[Efficacy and safety of long-acting carteolol 1% once daily. A double-masked, randomized study]". J Fr Ophtalmol. 26 (2): 131–6. PMID 12660585.
This article is issued from Wikipedia - version of the 5/25/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.