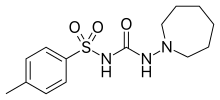
Tolazamide
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682482 |
| License data |
|
| Pregnancy category | |
| Routes of administration | Oral |
| ATC code | A10BB05 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | metabolized in the liver to active metabolites |
| Biological half-life | 7 hours |
| Excretion | Renal (85%) and fecal (7%) |
| Identifiers | |
| |
| CAS Number | 1156-19-0 |
| PubChem (CID) | 5503 |
| IUPHAR/BPS | 6847 |
| DrugBank |
DB00839 |
| ChemSpider |
5302 |
| UNII |
9LT1BRO48Q |
| KEGG |
D00379 |
| ChEBI | CHEBI:9613 |
| ChEMBL |
CHEMBL817 |
| ECHA InfoCard | 100.013.262 |
| Chemical and physical data | |
| Formula | C14H21N3O3S |
| Molar mass | 311.401 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Tolazamide is an oral blood glucose lowering drug used for people with Type 2 diabetes. It is part of the sulfonylurea family (ATC A10BB).
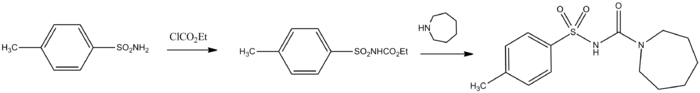
Synthesis
para-Toluenesulfonamide is converted to its carbamate with ethyl chloroformate in the presence of a base. Heating that intermediate with azepane leads to the displacement of the ethoxy group and the formation of tolazemide:[1]
References
External links
This article is issued from Wikipedia - version of the 8/24/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.