Tumor necrosis factor superfamily
| TNF (Tumor Necrosis Factor) family | |||||||||
|---|---|---|---|---|---|---|---|---|---|
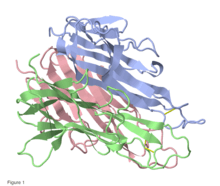 Trimeric structure of TNF alpha, produced by Mus musculus, based on PDB structure 2TNF (1.4 Å Resolution). Different colors represent different monomers. Baeyens, KJ et al. (1999).[1] Figure rendered using FirstGlance Jmol. | |||||||||
| Identifiers | |||||||||
| Symbol | TNF | ||||||||
| Pfam | PF00229 | ||||||||
| InterPro | IPR006052 | ||||||||
| PROSITE | PDOC00224 | ||||||||
| SCOP | 1tnf | ||||||||
| SUPERFAMILY | 1tnf | ||||||||
| OPM superfamily | 357 | ||||||||
| OPM protein | 2hew | ||||||||
| |||||||||
| TNF | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 crystal structure of trail-sdr5 | |||||||||
| Identifiers | |||||||||
| Symbol | TNF | ||||||||
| Pfam | PF00229 | ||||||||
| Pfam clan | CL0100 | ||||||||
| InterPro | IPR006052 | ||||||||
| PROSITE | PDOC00561 | ||||||||
| SCOP | 1tnr | ||||||||
| SUPERFAMILY | 1tnr | ||||||||
| |||||||||
The tumor necrosis factor (TNF) superfamily refers to a group of cytokines that can cause cell death (apoptosis).
The first two members of the family to be identified were:
- Tumor necrosis factor (TNF), formerly known as TNFα or TNF alpha, is the best-known member of this class. TNF is a monocyte-derived cytotoxin that has been implicated in tumor regression, septic shock, and cachexia.[2][3] The protein is synthesized as a prohormone with an unusually long and atypical signal sequence, which is absent from the mature secreted cytokine.[4] A short hydrophobic stretch of amino acids serves to anchor the prohormone in lipid bilayers.[5] Both the mature protein and a partially processed form of the hormone can be secreted after cleavage of the propeptide.[5]
- Lymphotoxin-alpha, formerly known as Tumor necrosis factor-beta (TNF-β), is a cytokine that is inhibited by interleukin 10.[6]
Family members
Nineteen proteins have been identified as part of the TNF family on the basis of sequence, functional, and structural similarities.[7] They include:[8][9][10]
- Tumor necrosis factor (TNF) (also known as cachectin [11] or TNF alpha)[12][13] is a cytokine that has a wide variety of functions. It can cause cytolysis of certain tumor cell lines; it is involved in the induction of cachexia; it is a potent pyrogen, causing fever by direct action or by stimulation of interleukin-1 secretion; it can stimulate cell proliferation and induce cell differentiation under certain conditions.
- Lymphotoxin-alpha (LT-alpha) and lymphotoxin-beta (LT-beta), two related cytokines produced by lymphocytes that are cytotoxic for a wide range of tumor cells in vitro and in vivo.[14]
- T cell antigen gp39 (CD40L), a cytokine that seems to be important in B-cell development and activation.
- CD27L, a cytokine that plays a role in T-cell activation. It induces the proliferation of co-stimulated T cells and enhances the generation of cytolytic T cells.
- CD30L, a cytokine that induces proliferation of T cells.
- FASL, a cell surface protein involved in cell death.[15]
- 4-1BBL, an inducible T cell surface molecule that contributes to T-cell stimulation.
- OX40L, a cell surface protein that co-stimulates T cell proliferation and cytokine production.[16]
- TNF-related apoptosis inducing ligand (TRAIL), a cytokine that induces apoptosis.[17]
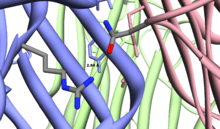
All these cytokines seem to form homotrimeric (or heterotrimeric in the case of LT-alpha/beta) complexes that are recognized by their specific receptors. Strong hydrogen bonds between the monomers stabilize the tertiary structure. One such example is the Asn34-Arg82 hydrogen bond in the M. musculus TNF alpha.[1] The PROSITE pattern for this family is located in a beta-strand in the central section of the protein that is conserved across all members.
All members of the TNF family, with the exception of the secreted lymphotoxin and a proliferation-inducing ligand (APRIL), are type II transmembrane proteins that protrude from immune cells. Such membrane-bound TNF ligands frequently signal back to the immune cells when they contact and bind their cognate receptors on other cells.[7]
Examples
Human proteins containing this domain include:
- CD40LG (TNFSF5); CD70 (TNFSF7); EDA; FASLG (TNFSF6); LTA (TNFSF1); LTB (TNFSF3);
- TNF
- TNFSF4 (OX40L); TNFSF8 (CD153); TNFSF9; TNFSF10 (TRAIL); TNFSF11 (RANKL); TNFSF12 (TWEAK); TNFSF13; TNFSF13B; TNFSF14; TNFSF15; TNFSF18;
Notes and references
This article incorporates text from the public domain Pfam and InterPro IPR006052
- 1 2 3 Baeyens KJ, De Bondt HL, Raeymaekers A, Fiers W, De Ranter CJ (April 1999). "The structure of mouse tumour-necrosis factor at 1.4 Å resolution: towards modulation of its selectivity and trimerization". Acta Crystallogr. D. 55 (Pt 4): 772–8. doi:10.1107/s0907444998018435. PMID 10089307.
- ↑ Fransen L, Müller R, Marmenout A, Tavernier J, Van der Heyden J, Kawashima E, Chollet A, Tizard R, Van Heuverswyn H, Van Vliet A (June 1985). "Molecular cloning of mouse tumour necrosis factor cDNA and its eukaryotic expression". Nucleic Acids Res. 13 (12): 4417–29. doi:10.1093/nar/13.12.4417. PMC 321797
 . PMID 2989794.
. PMID 2989794. - ↑ Kriegler M, Perez C, DeFay K, Albert I, Lu SD (April 1988). "A novel form of TNF/cachectin is a cell surface cytotoxic transmembrane protein: ramifications for the complex physiology of TNF". Cell. 53 (1): 45–53. doi:10.1016/0092-8674(88)90486-2. PMID 3349526.
- ↑ Sherry B, Jue DM, Zentella A, Cerami A (December 1990). "Characterization of high molecular weight glycosylated forms of murine tumor necrosis factor". Biochem. Biophys. Res. Commun. 173 (3): 1072–8. doi:10.1016/S0006-291X(05)80895-2. PMID 2268312.
- 1 2 Cseh K, Beutler B (September 1989). "Alternative cleavage of the cachectin/tumor necrosis factor propeptide results in a larger, inactive form of secreted protein". J. Biol. Chem. 264 (27): 16256–60. PMID 2777790.
- ↑ Waltenbaugh C, Doan T, Melvold R, Viselli S (2008). Immunology. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 68. ISBN 0-7817-9543-5.
- 1 2 Sun M, Fink PJ (2007). "A new class of reverse signaling costimulators belongs to the TNF family". J Immunol. 179 (7): 4307–12. doi:10.4049/jimmunol.179.7.4307. PMID 17878324.
- ↑ Peitsch MC, Jongeneel CV (February 1993). "A 3-D model for the CD40 ligand predicts that it is a compact trimer similar to the tumor necrosis factors". Int. Immunol. 5 (2): 233–8. doi:10.1093/intimm/5.2.233. PMID 8095800.
- ↑ Farrah T, Smith CA (July 1992). "Emerging cytokine family". Nature. 358 (6381): 26. doi:10.1038/358026b0. PMID 1377364.
- ↑ Bazan JF (September 1993). "Emerging families of cytokines and receptors". Curr. Biol. 3 (9): 603–6. doi:10.1016/0960-9822(93)90009-D. PMID 15335677.
- ↑ D. CAPUT, et al., Identification of a common nucleotide sequence in the 3'-untranslated region of mRNA molecules specifying inflammatory mediators, Proc. Natl. Acad. Sci. USA 83:1670-1674 Biochemistry, 1986 and references cited)
- ↑ Beutler B, Cerami A (October 1988). "The history, properties, and biological effects of cachectin". Biochemistry. 27 (20): 7575–82. doi:10.1021/bi00420a001. PMID 3061461.
- ↑ Vilcek J, Lee TH (April 1991). "Tumor necrosis factor. New insights into the molecular mechanisms of its multiple actions". J. Biol. Chem. 266 (12): 7313–6. PMID 1850405.
- ↑ Browning JL, Ngam-ek A, Lawton P, DeMarinis J, Tizard R, Chow EP, Hession C, O'Brine-Greco B, Foley SF, Ware CF (March 1993). "Lymphotoxin beta, a novel member of the TNF family that forms a heteromeric complex with lymphotoxin on the cell surface". Cell. 72 (6): 847–56. doi:10.1016/0092-8674(93)90574-A. PMID 7916655.
- ↑ Suda T, Takahashi T, Golstein P, Nagata S (December 1993). "Molecular cloning and expression of the Fas ligand, a novel member of the tumor necrosis factor family". Cell. 75 (6): 1169–78. doi:10.1016/0092-8674(93)90326-L. PMID 7505205.
- ↑ Baum PR, Gayle RB, Ramsdell F, Srinivasan S, Sorensen RA, Watson ML, Seldin MF, Baker E, Sutherland GR, Clifford KN (September 1994). "Molecular characterization of murine and human OX40/OX40 ligand systems: identification of a human OX40 ligand as the HTLV-1-regulated protein gp34". EMBO J. 13 (17): 3992–4001. PMC 395319
 . PMID 8076595.
. PMID 8076595. - ↑ Wiley SR, Schooley K, Smolak PJ, Din WS, Huang CP, Nicholl JK, Sutherland GR, Smith TD, Rauch C, Smith CA (December 1995). "Identification and characterization of a new member of the TNF family that induces apoptosis". Immunity. 3 (6): 673–82. doi:10.1016/1074-7613(95)90057-8. PMID 8777713.
External links
- Tumor Necrosis Factors at the US National Library of Medicine Medical Subject Headings (MeSH)
- pex1 tumor necrosis factor gene