Sitagliptin
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a606023 |
| License data |
|
| Pregnancy category | |
| Routes of administration | Oral |
| ATC code | A10BH01 (WHO) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 87% |
| Protein binding | 38% |
| Metabolism | Hepatic (CYP3A4- and CYP2C8-mediated) |
| Biological half-life | 8 to 14 h[1] |
| Excretion | Renal (80%)[1] |
| Identifiers | |
| |
| CAS Number |
486460-32-6 |
| PubChem (CID) | 4369359 |
| IUPHAR/BPS | 6286 |
| DrugBank |
DB01261 |
| ChemSpider |
3571948 |
| UNII |
QFP0P1DV7Z |
| ChEBI |
CHEBI:40237 |
| ChEMBL |
CHEMBL1422 |
| ECHA InfoCard | 100.217.948 |
| Chemical and physical data | |
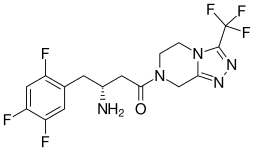
| Formula | C16H15F6N5O |
| Molar mass | 407.314 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
Sitagliptin (INN; ![]() i/sɪtəˈɡlɪptɪn/, previously identified as MK-0431 and marketed as the phosphate salt under the trade name Januvia) is an oral antihyperglycemic (antidiabetic drug) of the dipeptidyl peptidase-4 (DPP-4) inhibitor class. It was developed, and is marketed, by Merck & Co. This enzyme-inhibiting drug is used either alone or in combination with other oral antihyperglycemic agents (such as metformin or a thiazolidinedione) for treatment of diabetes mellitus type 2.[2]
i/sɪtəˈɡlɪptɪn/, previously identified as MK-0431 and marketed as the phosphate salt under the trade name Januvia) is an oral antihyperglycemic (antidiabetic drug) of the dipeptidyl peptidase-4 (DPP-4) inhibitor class. It was developed, and is marketed, by Merck & Co. This enzyme-inhibiting drug is used either alone or in combination with other oral antihyperglycemic agents (such as metformin or a thiazolidinedione) for treatment of diabetes mellitus type 2.[2]
Adverse effects
Side effects are as common with sitagliptin (whether used alone or with metformin or pioglitazone) as they were with placebo, except for rare nausea and common cold-like symptoms, including photosensitivity.[3] There is no significant difference in the occurrence of hypoglycemia between placebo and sitagliptin.[3][4][5] In those taking sulphonylureas there is an increased risk of low blood sugar.[6]
The existence of rare case reports of renal failure and hypersensitivity reactions is noted in the United States prescribing information, but a causative role for sitagliptin has not been established.[7]
There have been several postmarketing reports of pancreatitis (some fatal) in people treated with sitagliptin and other DPP-4 inhibitors,[8] and the U.S. package insert carries a warning to this effect,[9] although the causal link between sitagliptin and pancreatitis has not yet been fully substantiated.[2] One study with lab rats published in 2009 concluded that some of the possible risks of pancreatitis or pancreatic cancer may be reduced when it is used with metformin. However, while DPP-4 inhibitors showed an increase in such risk factors, as of 2009, there had been no reported increase in pancreatic cancer in individuals taking DPP-4 inhibitors.[10]
The updated (August 2015) prescribing information cautions that there have been multiple postmarketing reports of serious hypersensitivity reactions in patients receiving Sitagliptin. Merck notes that:
Additional adverse reactions have been identified during postapproval use of JANUVIA as monotherapy and/or in combination with other antihyperglycemic agents. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.Hypersensitivity reactions including anaphylaxis, angioedema, rash, urticaria, cutaneous vasculitis, and exfoliative skin conditions including Stevens-Johnson syndrome; hepatic enzyme elevations; acute pancreatitis, including fatal and non-fatal hemorrhagic and necrotizing pancreatitis; worsening renal function, including acute renal failure (sometimes requiring dialysis); severe and disabling arthralgia; constipation; vomiting; headache; myalgia; pain in extremity; back pain; pruritus; pemphigoid.[7]
In 2015, FDA added a new Warning and Precaution about the risk of "severe and disabling" joint pain to the labels of all DPP-4 Inhibitor medicines.[11] In addition to Sitagliptin, other DPP-4 inhibitors such as Saxagliptin, Linagliptin, and Alogliptin must also carry the new FDA Warning and Precaution label.
Mechanism of action
Sitagliptin works to competitively inhibit the enzyme dipeptidyl peptidase 4 (DPP-4). This enzyme breaks down the incretins GLP-1 and GIP, gastrointestinal hormones released in response to a meal.[12] By preventing GLP-1 and GIP inactivation, they are able to increase the secretion of insulin and suppress the release of glucagon by the alpha cells of the pancreas. This drives blood glucose levels towards normal. As the blood glucose level approaches normal, the amounts of insulin released and glucagon suppressed diminishes, thus tending to prevent an "overshoot" and subsequent low blood sugar (hypoglycemia) which is seen with some other oral hypoglycemic agents.
Sitagliptin has been shown to lower HbA1c level by about 0.7% points versus placebo. It is slightly less effective than metformin when used as a monotherapy. It does not cause weight gain and has less hypoglycemia compared to sulfonylureas. Sitagliptin is recommended as a second line drug (in combination with other drugs) after the combination of diet/exercise and metformin fails.[13]
History
Sitagliptin was approved by the U.S. Food and Drug Administration (FDA) on October 17, 2006,[14] and is marketed in the US as Januvia by Merck & Co. On April 2, 2007, the FDA approved an oral combination of sitagliptin and metformin marketed in the US as Janumet. On October 7, 2011, the FDA approved an oral combination of sitagliptin and simvastatin marketed in the US as Juvisync.[15]
See also
References
- 1 2 Herman GA, Stevens C, van Dyck K, Bergman A, Yi B, De Smet M, Snyder K, Hilliard D, Tanen M, Tanaka W, Wang AQ, Zeng W, Musson D, Winchell G, Davies MJ, Ramael S, Gottesdiener KM, Wagner JA (December 2005). "Pharmacokinetics and pharmacodynamics of sitagliptin, an inhibitor of dipeptidyl peptidase IV, in healthy subjects: results from two randomized, double-blind, placebo-controlled studies with single oral doses". Clin Pharmacol Ther. 78 (6): 675–88. doi:10.1016/j.clpt.2005.09.002. PMID 16338283.
- 1 2 National Prescribing Service (August 2010). "Sitagliptin for Type 2 Diabetes". Retrieved 27 August 2010.
- 1 2 "Januvia Side Effects & Drug Interactions". RxList.com. 2007. Retrieved 2007-11-28.
- ↑ http://dermatology.cdlib.org/1802/04_csp/09_11-00188/article.html
- ↑ http://www.ehealthme.com/ds/januvia/photosensitivity+reaction
- ↑ Salvo, Francesco; Moore, Nicholas; Arnaud, Mickael; Robinson, Philip; Raschi, Emanuel; De Ponti, Fabrizio; Bégaud, Bernard; Pariente, Antoine (3 May 2016). "Addition of dipeptidyl peptidase-4 inhibitors to sulphonylureas and risk of hypoglycaemia: systematic review and meta-analysis". BMJ. 353: i2231. doi:10.1136/bmj.i2231. PMC 4854021
 . PMID 27142267.
. PMID 27142267. - 1 2 "www.merck.com" (PDF).
- ↑ Olansky L (2010). "Do incretin-based therapies cause acute pancreatitis?". J Diabetes Sci Technol. 4 (1): 228–9. doi:10.1177/193229681000400129. PMC 2825646
 . PMID 20167189.
. PMID 20167189. - ↑ "Januvia for type 2 diabetes". Merck & Co. Retrieved 2010-07-31.
- ↑ Aleksey V. Matveyenko, Sarah Dry, Heather I. Cox, Artemis Moshtaghian1, Tatyana Gurlo, Ryan Galasso, Alexandra E. Butler and Peter C. Butler, Beneficial Endocrine but Adverse Exocrine Effects of Sitagliptin in the Human Islet Amyloid Polypeptide Transgenic Rat Model of Type 2 Diabetes—Interactions With Metformin Diabetes July 2009 vol. 58 no. 7 1604–1615
- ↑ "DPP-4 Inhibitors for Type 2 Diabetes: Drug Safety Communication—May Cause Severe Joint Pain". FDA. 2015-08-28. Retrieved 1 September 2015.
- ↑ Herman G, Bergman A, Liu F, Stevens C, Wang A, Zeng W, Chen L, Snyder K, Hilliard D, Tanen M, Tanaka W, Meehan A, Lasseter K, Dilzer S, Blum R, Wagner J (2006). "Pharmacokinetics and pharmacodynamic effects of the oral DPP-4 inhibitor sitagliptin in middle-aged obese subjects.". J Clin Pharmacol. 46 (8): 876–86. doi:10.1177/0091270006289850. PMID 16855072.
- ↑ Gadsby, Roger (2009). "Efficacy and Safety of Sitagliptin in the Treatment of Type 2 Diabetes" (pdf). Clinical Medicine: Therapeutics (1): 53–62.
- ↑ "FDA Approves New Treatment for Diabetes" (Press release). U.S. Food and Drug Administration (FDA). October 17, 2006. Retrieved 2006-10-17.
- ↑ "FDA Approves Combination Therapy Juvisync" (Press release). U.S. Food and Drug Administration (FDA). October 7, 2011. Retrieved 2013-11-17.
External links
- Official website
- Januvia Prescribing Information – Merck & Co.
- Sitagliptin Pharmacokinetics – PubPK
- Merck Announces FDA Acceptance of New Drug Application for JANUVIA – Merck press release.
- The race to get DPP-4 inhibitors to market at Archive.is (archived 2012-12-06) – Forbes.com
- Merck's March Madness – Forbes.com
- Banting and Best Diabetes Centre at UT sitagliptin – Sitagliptin
- Banting and Best Diabetes Centre at UT dpp4 – About DPP-4
- U.S. National Library of Medicine: Drug Information Portal – Sitagliptin