List of antioxidants in food
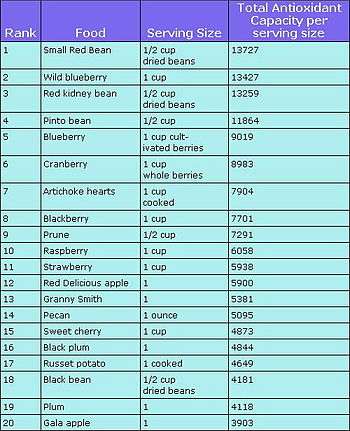
Food sources
- Main article: oxygen radical absorbance capacity
Regulatory guidance
In the following discussion, the term "antioxidant" refers mainly to non-nutrient compounds in foods, such as polyphenols, which have antioxidant capacity in vitro and so provide an artificial index of antioxidant strength – the ORAC measurement. Other than for dietary antioxidant vitamins – vitamin A, vitamin C and vitamin E – no food compounds have been proved with antioxidant efficacy in vivo. Accordingly, regulatory agencies like the Food and Drug Administration of the United States and the European Food Safety Authority (EFSA) have published guidance disallowing food product labels to claim an inferred antioxidant benefit when no such physiological evidence exists.[1][2]
Polyphenols in foods
Many common foods contain rich sources of polyphenols which have antioxidant properties only in test tube studies. As interpreted by the Linus Pauling Institute, dietary polyphenols have little or no direct antioxidant food value following digestion.[3] Not like controlled test tube conditions, the fate of flavones or polyphenols in vivo shows they are poorly conserved (less than 5%), with most of what is absorbed existing as metabolites modified during digestion and destined for rapid excretion.[4]
Spices, herbs, and essential oils are rich in polyphenols in the plant itself and shown with antioxidant potential in vitro. Typical spices high in polyphenols (confirmed in vitro) are clove, cinnamon, oregano, turmeric, cumin, parsley, basil, curry powder, mustard seed, ginger, pepper, chili powder, paprika, garlic, coriander, onion and cardamom. Typical herbs are sage, thyme, marjoram, tarragon, peppermint, oregano, savory, basil and dill weed.
Dried fruits are a good source of polyphenols by weight/serving size as the water has been removed making the ratio of polyphenols higher. Typical dried fruits are pears, apples, plums, peaches, raisins, figs and dates. Dried raisins are high in polyphenol count. Red wine is high in total polyphenol count which supplies antioxidant quality which is unlikely to be conserved following digestion (see section below).
Deeply pigmented fruits like cranberries, blueberries, plums, blackberries, raspberries, strawberries, blackcurrants, figs, cherries, guava, oranges, mango, grape juice and pomegranate juice also have significant polyphenol content.
Typical cooked vegetables rich in antioxidants are artichokes, cabbage, broccoli, asparagus, avocados, beetroot and spinach.
Nuts are a moderate source of polyphenol antioxidants. Typical nuts are pecans, walnuts, hazelnuts, pistachio, almonds, cashew nuts, macadamia nuts and peanut butter.
Sorghum bran, cocoa powder, and cinnamon are rich sources of procyanidins, which are large molecular weight compounds found in many fruits and some vegetables. Partly due to the large molecular weight (size) of these compounds, their amount actually absorbed in the body is low, an effect also resulting from the action of stomach acids, enzymes and bacteria in the gastrointestinal tract where smaller derivatives are metabolized and prepared for rapid excretion.[3][4]
Physiological context
Despite the above discussion implying that ORAC-rich foods with polyphenols may provide antioxidant benefits when in the diet, there remains no physiological evidence that any polyphenols have such actions or that ORAC has any relevance in the human body.
On the contrary, research indicates that although polyphenols are good antioxidants in vitro, antioxidant effects in vivo are probably negligible or absent.[5][6][7] By non-antioxidant mechanisms still undefined, polyphenols may affect mechanisms of cardiovascular disease or cancer.[8]
The increase in antioxidant capacity of blood seen after the consumption of polyphenol-rich (ORAC-rich) foods is not caused directly by the polyphenols, but most likely results from increased uric acid levels derived from metabolism of flavonoids.[3][4] According to Frei, "we can now follow the activity of flavonoids in the body, and one thing that is clear is that the body sees them as foreign compounds and is trying to get rid of them."[4] Another mechanism may be the increase in activities of paraoxonases by dietary antioxidants which can reduce oxidative stress.[9]
Vitamins
- Vitamin A (retinol), also synthesized by the body from beta-carotene, protects dark green, yellow and orange vegetables and fruits from solar radiation damage, and is thought to play a similar role in the human body. Carrots, squash, broccoli, sweet potatoes, tomatoes (which gain their color from the compound lycopene), kale, mangoes, oranges, seabuckthorn berries, wolfberries (goji), collards, cantaloupe, peaches and apricots are particularly rich sources of beta-carotene, the major provitamin A carotenoid.
- Vitamin C (ascorbic acid) is a water-soluble compound that fulfills several roles in living systems. Important sources include citrus fruits (such as oranges, sweet lime, etc.), green peppers, broccoli, green leafy vegetables, black currants, strawberries, blueberries, seabuckthorn, raw cabbage and tomatoes. Linus Pauling was a major advocate for its use.
- Vitamin E, including tocotrienol and tocopherol, is fat soluble and protects lipids. Sources include wheat germ, seabuckthorn, nuts, seeds, whole grains, green leafy vegetables, kiwifruit, vegetable oil, and fish-liver oil. Alpha-tocopherol is the main form in which vitamin E is consumed. Recent studies showed that some tocotrienol isomers have significant anti-oxidant properties.
Vitamin cofactors and minerals
- Coenzyme Q10
- Manganese, particularly when in its +2 valence state as part of the enzyme called superoxide dismutase (SOD).
- Iodide
Hormones
Carotenoid terpenoids
- Main article: carotenoid
- Alpha-carotene - found in carrots, winter squash, tomatoes, green beans, cilantro, Swiss chard
- Astaxanthin - found naturally in red algae and animals higher in the marine food chain. It is a red pigment familiarly recognized in crustacean shells and salmon flesh/roe.
- Beta-carotene - found in high concentrations in butternut squash, carrots, orange bell peppers, pumpkins, kale, peaches, apricots, mango, turnip greens, broccoli, spinach, and sweet potatoes.
- Canthaxanthin
- Lutein - found in high concentration in spinach, kale, Swiss chard, collard greens, beet and mustard greens, endive, red pepper and okra
- Lycopene - found in high concentration in cooked red tomato products like canned tomatoes, tomato sauce, tomato juice and garden cocktails, guava and watermelons.
- Zeaxanthin - best sources are kale, collard greens, spinach, turnip greens, Swiss chard, mustard and beet greens, corn, and broccoli
Natural phenols
Natural phenols are a class of molecules found in abundance in plants.
Flavonoids
Flavonoids, a subset of polyphenol antioxidants, are present in many berries, as well as in coffee and tea.
- Flavones:
- Flavonols:
- Isorhamnetin
- Kaempferol
- Myricetin - walnuts are a rich source
- Proanthocyanidins, or condensed tannins
- Quercetin and related, such as rutin
- Flavanones:
- Eriodictyol
- Hesperetin (metabolizes to hesperidin)
- Naringenin (metabolized from naringin)
- Flavanols and their polymers:
- Catechin, gallocatechin and their corresponding gallate esters
- Epicatechin, epigallocatechin and their corresponding gallate esters
- Theaflavin its gallate esters
- Thearubigins
- Isoflavone phytoestrogens - found primarily in soy, peanuts, and other members of the Fabaceae family
- Stilbenoids:
- Resveratrol - found in the skins of dark-colored grapes, and concentrated in red wine.
- Pterostilbene - methoxylated analogue of resveratrol, abundant in Vaccinium berries
- Anthocyanins
Phenolic acids and their esters
- Main article: polyphenol antioxidant
- Chicoric acid - another caffeic acid derivative, is found in chicory and Echinacea.
- Chlorogenic acid - found in high concentration in coffee (more concentrated in robusta than arabica beans), blueberries and tomatoes. Produced from esterification of caffeic acid.
- Cinnamic acid and its derivatives, such as ferulic acid - found in seeds of plants such as in brown rice, whole wheat and oats, as well as in coffee, apple, artichoke, peanut, orange and pineapple.
- Ellagic acid - found in high concentration in raspberry and strawberry, and in ester form in red wine tannins.
- Ellagitannins - hydrolyzable tannin polymer formed when ellagic acid, a polyphenol monomer, esterifies and binds with the hydroxyl group of a polyol carbohydrate such as glucose.
- Gallic acid - found in gallnuts, sumac, witch hazel, tea leaves, oak bark, and many other plants.
- Gallotannins - hydrolyzable tannin polymer formed when gallic acid, a polyphenol monomer, esterifies and binds with the hydroxyl group of a polyol carbohydrate such as glucose.
- Rosmarinic acid - found in high concentration in rosemary, oregano, lemon balm, sage, and marjoram.
- Salicylic acid - found in most vegetables, fruits, and herbs; but most abundantly in the bark of willow trees, from where it was extracted for use in the early manufacture of aspirin.
Other nonflavonoid phenolics
- Curcumin - Curcumin has low bioavailability, because, much of it is excreted through glucuronidation. However, bioavailability is substantially enhanced by solubilization in a lipid (oil or lecithin),[10] heat,[11] addition of piperine,[10] or through nanoparticularization.[12]
- Flavonolignans - e.g. silymarin - a mixture of flavonolignans extracted from milk thistle.
- Xanthones - mangosteen is purported to contain a large variety of xanthones,[13] but some of the xanthones like mangostin might be present only in the inedible shell.
- Eugenol
Other potential organic antioxidants
- Capsaicin, the active component of chili peppers
- Bilirubin, a breakdown product of blood, has been identified as a possible antioxidant.[14]
- Citric acid, oxalic acid, and phytic acid
- N-Acetylcysteine, water-soluble
- R-α-Lipoic acid, fat- and water-soluble
- Uric acid, in humans, accounts for roughly half the antioxidant capacity of plasma. Fructose, which is found abundantly in fruits, significantly elevates uric acid levels in humans,[15] and thus indirectly increases antioxidant capacity. High levels of uric acid may be a protective factor against Parkinson's disease[16] and possibly other diseases related to oxidative stress.
See also
References
- ↑ Guidance for Industry, Food Labeling; Nutrient Content Claims; Definition for "High Potency" and Definition for "Antioxidant" for Use in Nutrient Content Claims for Dietary Supplements and Conventional Foods U.S. Department of Health and Human Services, Food and Drug Administration, Center for Food Safety and Applied Nutrition, June 2008
- ↑ EFSA Panel on Dietetic Products, Nutrition and Allergies (2010). "Scientific Opinion on the substantiation of health claims related to various food(s)/food constituent(s) and protection of cells from premature aging, antioxidant activity, antioxidant content and antioxidant properties, and protection of DNA, proteins and lipids from oxidative damage pursuant to Article 13(1) of Regulation (EC) No 1924/2006". EFSA Journal. 8 (2): 1489. doi:10.2903/j.efsa.2010.1489.
- 1 2 3 Lotito, S; Frei, B (2006). "Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon?". Free Radical Biology and Medicine. 41 (12): 1727–46. doi:10.1016/j.freeradbiomed.2006.04.033. PMID 17157175.
- 1 2 3 4 David Stauth (5 March 2007). "Studies force new view on biology of flavonoids". EurekAlert!; Adapted from a news release issued by Oregon State University.
- ↑ Williams, Robert J; Spencer, Jeremy P.E; Rice-Evans, Catherine (2004). "Flavonoids: antioxidants or signalling molecules?☆". Free Radical Biology and Medicine. 36 (7): 838–49. doi:10.1016/j.freeradbiomed.2004.01.001. PMID 15019969.
- ↑ Gross, P (2009). "New Roles for Polyphenols. A 3-Part report on Current Regulations & the State of Science". Nutraceuticals World. Rodman Media. Retrieved April 11, 2013.
- ↑ Jonny Bowden, PhD, C.N.S. (16 Dec 2012). "ORAC no more!". Huffington Post. Retrieved 12 Dec 2012.
- ↑ Arts, IC; Hollman, PC (2005). "Polyphenols and disease risk in epidemiologic studies". The American Journal of Clinical Nutrition. 81 (1 Suppl): 317S–325S. PMID 15640497.
- ↑ Aviram, M; Rosenblat, M (2005). "Paraoxonases and cardiovascular diseases: pharmacological and nutritional influences". Current Opinion in Lipidology. 16 (4): 393–9. doi:10.1097/01.mol.0000174398.84185.0f. PMID 15990587.
- 1 2 Anand, Preetha; Kunnumakkara, Ajaikumar B.; Newman, Robert A.; Aggarwal, Bharat B. (2007). "Bioavailability of Curcumin: Problems and Promises". Molecular Pharmaceutics. 4 (6): 807–18. doi:10.1021/mp700113r. PMID 17999464.
- ↑ Kurien, Biji T.; Singh, Anil; Matsumoto, Hiroyuki; Scofield, R. Hal (2007). "Improving the Solubility and Pharmacological Efficacy of Curcumin by Heat Treatment". ASSAY and Drug Development Technologies. 5 (4): 567–76. doi:10.1089/adt.2007.064. PMID 17767425.
- ↑ Nair, Hareesh B.; Sung, Bokyung; Yadav, Vivek R.; Kannappan, Ramaswamy; Chaturvedi, Madan M.; Aggarwal, Bharat B. (2010). "Delivery of antiinflammatory nutraceuticals by nanoparticles for the prevention and treatment of cancer". Biochemical Pharmacology. 80 (12): 1833–1843. doi:10.1016/j.bcp.2010.07.021. PMC 2974020
 . PMID 20654584.
. PMID 20654584. - ↑ Archived August 4, 2008, at the Wayback Machine.
- ↑ Stocker, R; Yamamoto, Y; McDonagh, A.; Glazer, A.; Ames, B. (1987). "Bilirubin is an antioxidant of possible physiological importance". Science. 235 (4792): 1043–6. Bibcode:1987Sci...235.1043S. doi:10.1126/science.3029864. PMID 3029864.
- ↑ Zawiasa, A.; Szklarek-Kubicka, M.; Fijałkowska-Morawska, J.; Nowak, D.; Rysz, J.; Mamełka, B.; Nowicki, M. (2009). "Effect of Oral Fructose Load on Serum Uric Acid and Lipids in Kidney Transplant Recipients Treated with Cyclosporine or Tacrolimus". Transplantation Proceedings. 41 (1): 188–91. doi:10.1016/j.transproceed.2008.10.038. PMID 19249511.
- ↑ De Vera, Mary; Rahman, M. Mushfiqur; Rankin, James; Kopec, Jacek; Gao, Xiang; Choi, Hyon (2008). "Gout and the risk of parkinson's disease: A cohort study". Arthritis & Rheumatism. 59 (11): 1549–54. doi:10.1002/art.24193. PMID 18975349.
External links
- The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide Nutr J. 2010