Porfimer sodium
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Consumer Drug Information |
| License data | |
| Pregnancy category |
|
| Routes of administration | Intravenous |
| ATC code | L01XD01 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | NA |
| Protein binding | ~90% |
| Biological half-life | 21.5 days (mean) |
| Excretion | Fecal |
| Identifiers | |
| CAS Number |
97067-70-4 |
| PubChem (CID) | 57166 |
| DrugBank |
DB00707 |
| ChemSpider |
10482283 |
| UNII |
Y3834SIK5F |
| ChEMBL |
CHEMBL1201707 |
| Chemical and physical data | |
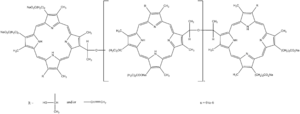
| Formula | C68H74N8O11 (for n=0) |
| Molar mass | 1179.36 g/mol (for n=0) |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Porfimer sodium, sold as Photofrin, is a photosensitizer used in photodynamic therapy and radiation therapy and for palliative treatment of obstructing endobronchial non-small cell lung carcinoma and obstructing esophageal cancer.
Porfimer is a mixture of oligomers formed by ether and ester linkages of up to eight porphyrin units.[1] In practice, a red light source emitting at 630 nm is used to excite the Porfimer oligomers.[2]
Porfimer is Haematoporphyrin Derivative (HpD) (See PDT).
Approvals and indications
It was approved in Canada in 1993 for the treatment of bladder cancer.[2] It was approved in Japan in 1994 (for early stage lung cancer?).[2] It was approved by the U.S. FDA in December 1995 for esophageal cancer, and in 1998, it was approved for the treatment of early non-small cell lung cancer.[2]
In August 2003 the FDA approved its use for Barrett's esophagus.[3]
References
- ↑ "Porfimer injection Prescribing information" (PDF).
- 1 2 3 4 "Photodynamic Therapy (PDT) for Lung Cancers". 2006.
- ↑ "FDA Patient Safety News: Show #20, October 2003". October 2003. Retrieved 2009-08-17.