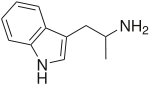
α-Methyltryptamine
 |
| Clinical data |
|---|
Routes of
administration |
Oral, Insufflation, Rectal, Smoked, IM, IV[1] |
|---|
| ATC code |
none |
|---|
| Legal status |
|---|
| Legal status |
|
|---|
| Identifiers |
|---|
- 1-(1H-Indol-3-yl)propan-2-amine
|
| Synonyms |
Indopan; IT-290, IT-403, U-14,164E, 3-IT[1] |
|---|
| CAS Number |
299-26-3  Y Y |
|---|
| PubChem (CID) |
9287 |
|---|
| DrugBank |
DB01446  Y Y |
|---|
| ChemSpider |
8930  Y Y |
|---|
| ChEBI |
CHEBI:59020  Y Y |
|---|
| ChEMBL |
CHEMBL30713  Y Y |
|---|
| Chemical and physical data |
|---|
| Formula |
C11H14N2 |
|---|
| Molar mass |
174.24 g/mol |
|---|
| 3D model (Jmol) |
Interactive image |
|---|
|
|
InChI=1S/C11H14N2/c1-8(12)6-9-7-13-11-5-3-2-4-10(9)11/h2-5,7-8,13H,6,12H2,1H3  Y YKey:QSQQQURBVYWZKJ-UHFFFAOYSA-N  Y Y
|
| (verify) |
|---|
α-Methyltryptamine (abbreviated as αMT, AMT) is a psychedelic, stimulant, and entactogen drug of the tryptamine class.[2][3] It was originally developed as an antidepressant by workers at Upjohn in the 1960s,[4] and was used briefly as an antidepressant in Russia under the trade name Indopan before being discontinued.[5][6][7]
Chemistry
αMT is tryptamine with a methyl substituent at the alpha carbon. Its chemical relation to tryptamine is analogous to that of amphetamine to phenethylamine, amphetamine being α-methylphenethylamine. αMT is closely related to the neurotransmitter serotonin (5-hydroxytryptamine) which partially explains its mechanism of action.
Pharmacology
αMT acts as a relatively balanced reuptake inhibitor and releasing agent of the main three monoamines; serotonin, norepinephrine, and dopamine,[8] and as a non-selective serotonin receptor agonist.[9]
MAOI activity
αMT has been shown as a reversible inhibitor of the enzyme monoamine oxidase (MAO) in-vitro[10] and in-vivo.[11]
In rats the potency of αMT as an MAO-A inhibitor in the brain was approximately equal to that of harmaline at equimolar doses.[note 1] Dexamphetamine did not enhance the 5-hydroxytryptophan-induced rise of serotonin at any level.[12]
A typical dose of harmaline for MAO inhibition is 150 mg,[13] higher than any typical αMT dose[14] so αMT's MAOI activity at typical doses will be significant but not total. The danger rises exponentially as αMT doses approach 150 mg due to increased monoamine release and increased MAO inhibition.
Metabolism
2-Oxo-αMT, 6-hydroxy-αMT,[15] 7-hydroxy-αMT and 1′-hydroxy-αMT were detected as metabolites of αMT in male Wistar rats
.[16]
Dosage and effects
With 20–30 milligrams, euphoria, empathy, and psychedelic effects become apparent and can last as long as 12 hours.[17] A dose exceeding 40 mg is generally considered as strong. In rare cases or extreme doses the duration of effects might exceed 24 hours. αMT in freebase form is reported by users to have been smoked, with doses of between and 2–5 milligrams being cited.[1][2]
Reported side effects include anxiety, restlessness, muscle tension, jaw tightness, pupil dilation, tachycardia, headaches, nausea, and vomiting, among other effects that might commonly be attributed to LSD, psilocybin, DMT and MDMA, such as open-eye visuals, closed eye visuals and an altered state of mind.[2][18]
In spite of some reported experiential similarities to MDMA, the chemicals are structurally unrelated; αMT is a tryptamine while MDMA is a phenethylamine.
Like many other serotonin releasing agents, αMT's analogue αET has been shown to produce long-lasting serotonergic neurotoxicity at very high doses.[19] It is possible that αMT could cause the same neurotoxicity.
Legality
Australia
The 5-Methoxy analogue, 5-MeO-αMT is schedule 9 in Australia and αMT would be controlled as an analogue of this.[20]
China
As of October 2015 αMT is a controlled substance in China.[21]
Denmark
In Denmark (2010), the Danish Minister for the Interior and Health placed αMT to their lists of controlled substances(List B).[22]
Canada
Canada has no mention of αMT in the Controlled Drugs and Substances Act.[23] However, αMT is on the Registration, Evaluation, Authorisation and Restriction of Chemicals(REACH) list of pre-registered substances as of March 2009.[22]
Germany
αMT is listed under the Narcotics Act in schedule 1 (narcotics not eligible for trade and medical prescriptions) in Germany.[22]
Austria
αMT is placed under Austrian law (NPSG) Group 6.[22]
Hungary
αMT was controlled on the Schedule C list in Hungary in 2013.[22]
Slovakia
αMT was placed in 2013 on the List of Hazardous Substances in Annex, § 2 in Slovakia.[22]
Slovenia
αMT appeared on the Decree on Classification of Illicit Drugs in Slovenia (2013).[22]
Lithuania
In Lithuania (2012), αMT is controlled as a tryptamine derivative put under control in the 1st list of Narcotic Drugs and Psychotropic Substances which use is prohibited for medical purposes.[22]
Spain
αMT is legal in Spain.[24]
Sweden
Sveriges riksdags health ministry Statens folkhälsoinstitut classified αMT as "health hazard" under the act Lagen om förbud mot vissa hälsofarliga varor (translated Act on the Prohibition of Certain Goods Dangerous to Health) as of Mar 1, 2005, in their regulation SFS 2005:26 listed as alfa-metyltryptamin (AMT), making it illegal to sell or possess.[25]
UK
αMT was made illegal in the United Kingdom as of 7 January 2015, along with 5-MeO-DALT.[26]
This was following the events of 10 June 2014 when the Advisory Council on the Misuse of Drugs recommended that αMT be scheduled as a class A drug by updating the blanket ban clause on tryptamines.[27]
USA
The Drug Enforcement Administration (DEA) placed αMT temporarily in schedule I of the Controlled Substances Act (CSA) on April 4, 2003, pursuant to the temporary scheduling provisions of the CSA (68 FR16427). On September 29, 2004, αMT was permanently controlled as a schedule I substance under the CSA (69FR 58050).[28]
Finland
AMT, alfa-methyltryptamine is illegal in Finland[29]
Reported Deaths
Deaths from αMT are rare but as a powerful monoamine releaser injury can occur when excessive doses are taken or when taken with drugs such as MAOIs.[30] Most fatalities are not verified but those which are involve excessive doses[31] or coingestion with other drugs.[32] A British teenager died after consuming 1 g of αMT in August 2013.[33]
Synthesis
Prepn:[34][35][36][37]
See also
Notes
- ↑ MAOI potency was comparable at 7 µM/kg, equivalent to 1.5mg/kg of Harmaline and 1.2mg/kg of αMT. At 70µM/kg αMT was a much less effective MAOI than harmaline.[12]
References
- 1 2 3 "Erowid AMT Vault : FAQ by Dialtonez".
- 1 2 3 "Erowid Online Books : "TIHKAL" - #48 a-MT".
- ↑ "Erowid AMT (alpha-methyltryptamine) Vault".
- ↑ US Patent 3296072, Szmuszkovicz Jacob, "Method of Treating Mental Depression", published 1967-01-03, assigned to Upjohn Co
- ↑ Donald G. Barceloux (20 March 2012). Medical Toxicology of Drug Abuse: Synthesized Chemicals and Psychoactive Plants. John Wiley & Sons. pp. 196–. ISBN 978-0-471-72760-6.
- ↑ Leslie Iversen (11 November 2013). Handbook of Psychopharmacology: Volume 14 Affective Disorders: Drug Actions in Animals and Man. Springer Science & Business Media. pp. 132–. ISBN 978-1-4613-4045-4.
- ↑ Biological Research on Addiction: Comprehensive Addictive Behaviors and Disorders. Academic Press. 17 May 2013. pp. 632–. ISBN 978-0-12-398360-2.
- ↑ Nagai F, Nonaka R, Satoh Hisashi Kamimura K (March 2007). "The effects of non-medically used psychoactive drugs on monoamine neurotransmission in rat brain". European Journal of Pharmacology. 559 (2-3): 132–7. doi:10.1016/j.ejphar.2006.11.075. PMID 17223101.
- ↑ Nonaka R, Nagai F, Ogata A, Satoh K (December 2007). "In vitro screening of psychoactive drugs by [(35)S]GTPgammaS binding in rat brain membranes". Biological & Pharmaceutical Bulletin. 30 (12): 2328–33. doi:10.1248/bpb.30.2328. PMID 18057721.
- ↑ Arai, Y.; Toyoshima, Y.; Kinemuchi, H. (1986). "Studies of monoamine oxidase and semicarbazide-sensitive amine oxidase. II. Inhibition by .ALPHA.-methylated substrate-analogue monoamines, .ALPHA.-methyltryptamine, .ALPHA.-methylbenzylamine and two enantiomers of .ALPHA.-methylbenzylamine". The Japanese Journal of Pharmacology. 41 (2): 191–197. doi:10.1254/jjp.41.191.
- ↑ Greig, M. E.; Walk, R. A.; Gibbons, A. J. (1959). "The effect of three tryptamine derivatives on serotonin metabolism in vitro and in vivo". The Journal of Pharmacology and Experimental Therapeutics. 127: 110–115. PMID 13851725.
- 1 2 Gey, K. F.; Pletscher, A. (1962). "Effect of alpha-alkylated tryptamine derivatives on 5-hydroxytryptamine metabolism in vivo". British journal of pharmacology and chemotherapy. 19 (1): 161–167. doi:10.1111/j.1476-5381.1962.tb01437.x. PMC 1482243
 . PMID 13898151.
. PMID 13898151. - ↑ "Shulgin, A (1997) TIHKAL". Erowid.org. Retrieved 2013-10-16.
- ↑ "AMT Dosage". Erowid. 2011-02-02. Retrieved 2013-10-16.
- ↑ Szara, S. (1961). "6-Hydroxylation: An important metabolic route for α-methyltryptamine". Experientia. 17 (2): 76–76. doi:10.1007/BF02171429.
- ↑ Kanamori, T.; Kuwayama, K.; Tsujikawa, K.; Miyaguchi, H.; Iwata, Y. T.; Inoue, H. (2008). "In vivometabolism of α-methyltryptamine in rats: Identification of urinary metabolites". Xenobiotica. 38 (12): 1476–1486. doi:10.1080/00498250802491654. PMID 18982537.
- ↑ Wilcox, J. (2012). "Psychoactive Properties of Alpha-Methyltryptamine: Analysis from Self Reports of Users". Journal of Psychoactive Drugs. 44 (3): 274–276. doi:10.1080/02791072.2012.704592. PMID 23061328.
- ↑ "Erowid AMT Vault : Effects".
- ↑ Huang XM, Johnson MP, Nichols DE (July 1991). "Reduction in brain serotonin markers by alpha-ethyltryptamine (Monase)". European Journal of Pharmacology. 200 (1): 187–90. doi:10.1016/0014-2999(91)90686-K. PMID 1722753.
- ↑ "Erowid 5-MeO-AMT Vault : Legal Status".
- ↑ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. 27 September 2015. Retrieved 1 October 2015.
- 1 2 3 4 5 6 7 8 http://www.who.int/medicines/areas/quality_safety/4_20_Review.pdf
- ↑ "CSDA".
- ↑ http://www.aemps.gob.es/medicamentosUsoHumano/estupefacientesPsicotropos/home.htm
- ↑ "Förordning om ändring i förordningen (1999:58) om förbud mot vissa hälsofarliga varor;" (pdf), Lagen om förbud mot vissa hälsofarliga varor - SFS 2005:26 (in Swedish), February 2005, retrieved 2013-10-07
- ↑
- ↑ ACMD (10 June 2014). "Update of the Generic Definition for Tryptamines" (PDF). UK Home Office. p. 12. Retrieved 10 June 2014.
- ↑ "ALPHA-METHYLTRYPTAMINE" (PDF). DEA Office of Diversion Control. April 2013. Archived from the original on 2013-10-17. Retrieved 2013-10-10.
- ↑ http://www.finlex.fi/data/sdliite/liite/6194.pdf
- ↑ Gillman, P. K. (2005). "Monoamine oxidase inhibitors, opioid analgesics and serotonin toxicity". British Journal of Anaesthesia. 95 (4): 434–441. doi:10.1093/bja/aei210. PMID 16051647. "drugs such as MDMA, ecstasy (3,4-methylenedioxymethamphetamine), if combined with MAOIs (including moclobemide) do also cause fatalities because they act as serotonin releasers"
- ↑ Boland, Diane M.; Andollo W; Hime GW; Hearn WL (July–August 2005). "Fatality Due to Acute Alpha-methyltryptamine Intoxication" (PDF). Journal of Analytical Toxicology. 29: 394–397. doi:10.1093/jat/29.5.394. Retrieved October 1, 2013.
- ↑ "Call for ban on drug after reveller's death". Western Gazette. March 22, 2012. Archived from the original on 2013-10-16. Retrieved October 1, 2013.
- ↑ "Southampton 'legal high' death deemed 'accidental'". BBC News. 2013-11-12. Retrieved 2013-11-19.
- ↑ Snyder, H. R.; Katz, Leon (1947). "The Alkylation of Aliphatic Nitro Compounds with Gramine. A New Synthesis of Derivatives of Tryptamine1,2". Journal of the American Chemical Society. 69 (12): 3140–3142. doi:10.1021/ja01204a061. ISSN 0002-7863.
- ↑ Ash, A. S. F.; Wragg, W. R. (1958). "790. Synthesis of the 3-2?-aminopropyl- and 3-2?-aminobutyl-derivatives of 5-hydroxyindole, and an alternative synthesis of 5-hydroxytryptamine". Journal of the Chemical Society (Resumed): 3887. doi:10.1039/jr9580003887. ISSN 0368-1769.
- ↑ Terzian, A. G.; Safrasbekian, R. R.; Sukasian, R. S.; Tatevosian, G. T. (15 November 1961). "Synthesis and some pharmacological properties of alpha-methyltryptamine and its 5-methoxy derivative". Experientia. 17: 493–4. doi:10.1007/bf02158615. PMID 13920395.
- ↑ Lloyd, David H.; Nichols, David E. (1986). "Nickel boride/hydrazine hydrate reduction of aromatic and aliphatic nitro compounds. Synthesis of 4-(benzyloxy)indole and .alpha.-alkyltryptamines". The Journal of Organic Chemistry. 51 (22): 4294–4295. doi:10.1021/jo00372a037. ISSN 0022-3263.
External links
|
|---|
|
Psychedelics
(5-HT2A
agonists) | | |
|---|
| | | |
|---|
| HOT-* | |
|---|
| 25*-NB*
(excludes FLY) | |
|---|
| Subst.
mescaline | |
|---|
| | |
|---|
| 3C-* | |
|---|
| 4C-* | |
|---|
| FLY | |
|---|
| | |
|---|
| Others | |
|---|
|
|---|
| | |
|---|
| | | |
|---|
| |
- 4,5-DHP-DMT
- 2,N,N-TMT
- 4-AcO-DMT
- 4-HO-5-MeO-DMT
- 4,N,N-TMT
- 4-Propionyloxy-DMT
- 5,6-diBr-DMT
- 5-AcO-DMT
- 5-Bromo-DMT
- 5-MeO-2,N,N-TMT
- 5-MeO-4,N,N-TMT
- 5-MeO-α,N,N-TMT
- 5-MeO-DMT
- 5-N,N-TMT
- 7,N,N-TMT
- α,N,N-TMT
- (Bufotenin) 5-HO-DMT
- DMT
- Norbaeocystin
- (Psilocin) 4-HO-DMT
- (Psilocybin) 4-PO-DMT
|
|---|
| | |
|---|
| | |
|---|
| | |
|---|
| | |
|---|
| | |
|---|
| | |
|---|
| | |
|---|
| Others | |
|---|
|
|---|
| Others | |
|---|
|
|---|
|
Dissociatives
(NMDAR
antagonists) | |
|---|
|
Deliriants
(mAChR
antagonists) | |
|---|
|
| Others | |
|---|
|
|---|
|
| Adamantanes | |
|---|
|
| Adenosine antagonists | |
|---|
|
| Alkylamines | |
|---|
|
| Ampakines | |
|---|
|
| Arylcyclohexylamines | |
|---|
|
| Benzazepines | |
|---|
|
| Cholinergics | |
|---|
|
| Convulsants | |
|---|
|
| Eugeroics | |
|---|
|
| Oxazolines | |
|---|
|
| Phenethylamines |
- 1-(4-Methylphenyl)-2-aminobutane
- 1-Phenyl-2-(piperidin-1-yl)pentan-3-one
- 1-Methylamino-1-(3,4-methylenedioxyphenyl)propane
- 2-Fuoroamphetamine
- 2-Fuoromethamphetamine
- 2-OH-PEA
- 2-Phenyl-3-aminobutane
- 2-Phenyl-3-methylaminobutane
- 2,3-MDA
- 3-Fuoroamphetamine
- 3-Fluoroethamphetamine
- 3-Fluoromethcathinone
- 3-Methoxyamphetamine
- 3-Methylamphetamine
- 3,4-DMMC
- 4-BMC
- 4-CMC
- 4-Ethylamphetamine
- 4-Fluoroamphetamine
- 4-Fluoromethamphetamine
- 4-MA
- 4-Methylbuphedrone
- 4-Methylcathinone
- 4-MMA
- 4-Methylpentedrone
- 4-MTA
- 6-FNE
- AL-1095
- Alfetamine
- a-Ethylphenethylamine
- Amfecloral
- Amfepentorex
- Amfepramone
- Amidephrine
- 2-Amino-1,2-dihydronaphthalene
- 2-Aminoindane
- 5-(2-Aminopropyl)indole
- 2-Aminotetralin
- Acridorex
- Amphetamine (Dextroamphetamine, Levoamphetamine)
- Amphetaminil
- Arbutamine
- β-Methylphenethylamine
- β-Phenylmethamphetamine
- Benfluorex
- Benzedrone
- Benzphetamine
- BDB
- BOH
- 3-Benzhydrylmorpholine
- BPAP
- Buphedrone
- Bupropion
- Butylone
- Camfetamine
- Cathine
- Cathinone
- Chlorphentermine
- Cilobamine
- Cinnamedrine
- Clenbuterol
- Clobenzorex
- Cloforex
- Clortermine
- Cypenamine
- D-Deprenyl
- Denopamine
- Dimethoxyamphetamine
- Dimethylamphetamine
- Dimethylcathinone
- Dobutamine
- DOPA (Dextrodopa, Levodopa)
- Dopamine
- Dopexamine
- Droxidopa
- EBDB
- Ephedrine
- Epinephrine
- Epinine
- Etafedrine
- Ethcathinone
- Ethylnorepinephrine
- Ethylone
- Etilamfetamine
- Etilefrine
- Famprofazone
- Fencamfamine
- Fencamine
- Fenethylline
- Fenfluramine (Dexfenfluramine, Levofenfluramine)
- Fenproporex
- Feprosidnine
- Flephedrone
- Fludorex
- Formetorex
- Furfenorex
- Gepefrine
- Hexapradol
- Hexedrone
- HMMA
- Hordenine
- 4-Hydroxyamphetamine
- 5-Iodo-2-aminoindane
- Ibopamine
- IMP
- Indanylamphetamine
- Iofetamine
- Isoetarine
- Isoethcathinone
- Isoprenaline
- L-Deprenyl (Selegiline)
- Lefetamine
- Lisdexamfetamine
- Lophophine
- MBDB
- MDA
- MDBU
- MDEA
- MDMA
- MDMPEA
- MDOH
- MDPR
- MDPEA
- Mefenorex
- Mephedrone
- Mephentermine
- Metanephrine
- Metaraminol
- Mesocarb
- Methamphetamine (Dextromethamphetamine, Levomethamphetamine)
- Methoxamine
- Methoxyphenamine
- MMA
- Methcathinone
- Methedrone
- Methoxyphenamine
- Methylenedioxycathinone
- Methylone
- Mexedrone
- MMDA
- MMDMA
- MMMA
- Morforex
- N,alpha-Diethylphenylethylamine
- N-Benzyl-1-phenethylamine
- N-Ethylbuphedrone
- N-Ethylhexedrone
- N,N-Dimethylphenethylamine
- Naphthylamphetamine
- Nisoxetine
- Norepinephrine
- Norfenefrine
- Norfenfluramine
- Normetanephrine
- L-Norpseudoephedrine
- Octopamine (drug)
- Orciprenaline
- Ortetamine
- Oxifentorex
- Oxilofrine
- PBA
- PCA
- PCMA
- PHA
- Pentorex
- Pentedrone
- Pentylone
- Phenatine
- Phenpromethamine
- Phentermine
- Phenylalanine
- Phenylephrine
- Phenylpropanolamine
- Pholedrine
- PIA
- PMA
- PMEA
- PMMA
- PPAP
- Phthalimidopropiophenone
- Prenylamine
- Propylamphetamine
- Pseudoephedrine
- Ropinirole
- Salbutamol (Levosalbutamol)
- Sibutramine
- Synephrine
- Theodrenaline
- Tiflorex
- Tranylcypromine
- Tyramine
- Tyrosine
- Xylopropamine
- Zylofuramine
|
|---|
|
| Phenylmorpholines | |
|---|
|
| Piperazines | |
|---|
|
| Piperidines | |
|---|
|
| Pyrrolidines | |
|---|
|
| Racetams | |
|---|
|
| Tropanes | |
|---|
|
| Tryptamines | |
|---|
|
| Others | |
|---|
|
|
|
|---|
|
| |
|---|
| | 5-HT1A |
- Agonists: 8-OH-DPAT
- Adatanserin
- Amphetamine
- Antidepressants (e.g., etoperidone, nefazodone, trazodone, vilazodone, vortioxetine)
- Atypical antipsychotics (e.g., aripiprazole, asenapine, clozapine, lurasidone, quetiapine, ziprasidone)
- Azapirones (e.g., buspirone, eptapirone, gepirone, perospirone, tandospirone)
- Bay R 1531
- Befiradol
- BMY-14802
- Cannabidiol
- Dimemebfe
- Dopamine
- Ebalzotan
- Eltoprazine
- Ergolines (e.g., bromocriptine, cabergoline, dihydroergotamine, ergotamine, lisuride, LSD, methylergometrine (methylergonovine), methysergide, pergolide)
- F-11461
- F-12826
- F-13714
- F-14679
- F-15063
- F-15599
- Flesinoxan
- Flibanserin
- Flumexadol
- Lesopitron
- LY-293284
- LY-301317
- mCPP
- MKC-242
- Naluzotan
- NBUMP
- Osemozotan
- Oxaflozane
- Pardoprunox
- Piclozotan
- Rauwolscine
- Repinotan
- Roxindole
- RU-24969
- S-14506
- S-14671
- S-15535
- Sarizotan
- Serotonin (5-HT)
- SSR-181507
- Sunepitron
- Tryptamines (e.g., 5-CT, 5-MeO-DMT, 5-MT, bufotenin, DMT, indorenate, N-Me-5-HT, psilocin, psilocybin)
- TGBA01AD
- U-92016A
- Urapidil
- Vilazodone
- Xaliproden
- Yohimbine
| |
- Antagonists: Atypical antipsychotics (e.g., iloperidone, risperidone, sertindole)
- AV965
- Beta blockers (e.g., alprenolol, cyanopindolol, iodocyanopindolol, oxprenolol, pindobind, pindolol, propranolol, tertatolol)
- BMY-7378
- CSP-2503
- Dotarizine
- Ergolines (e.g., metergoline)
- Flopropione
- GR-46611
- Isamoltane
- Lecozotan
- Mefway
- Metitepine (methiothepin)
- MIN-117 (WF-516)
- MPPF
- NAN-190
- Robalzotan
- S-15535
- SB-649915
- SDZ 216-525
- Spiperone
- Spiramide
- Spiroxatrine
- UH-301
- WAY-100135
- WAY-100635
- Xylamidine
| | |
|
|---|
| | 5-HT1B |
- Agonists: CGS-12066A
- CP-93129
- CP-94253
- CP-122,288
- CP-135807
- Eltoprazine
- Ergolines (e.g., bromocriptine, dihydroergotamine, ergotamine, methylergometrine (methylergonovine), methysergide, pergolide)
- mCPP
- RU-24969
- Serotonin (5-HT)
- Triptans (e.g., avitriptan, donitriptan, eletriptan, sumatriptan, zolmitriptan)
- TFMPP
- Tryptamines (e.g., 5-BT, 5-CT, 5-MT, DMT)
- Vortioxetine
| | | | |
|
|---|
| | 5-HT1D |
- Agonists: CP-122,288
- CP-135807
- CP-286601
- Ergolines (e.g., bromocriptine, cabergoline, dihydroergotamine, ergotamine, LSD, methysergide)
- GR-46611
- L-694247
- L-772405
- mCPP
- PNU-109291
- PNU-142633
- Serotonin (5-HT)
- TGBA01AD
- Triptans (e.g., almotriptan, avitriptan, donitriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, sumatriptan, zolmitriptan)
- Tryptamines (e.g., 5-BT, 5-CT, 5-Et-DMT, 5-MT, 5-(nonyloxy)tryptamine, DMT)
| | | | |
|
|---|
| | 5-HT1E | |
|---|
| | 5-HT1F | |
|---|
|
|
|
| |
|---|
| | 5-HT2A |
- Agonists: 25H/NB series (e.g., 25I-NBF, 25I-NBMD, 25I-NBOH, 25I-NBOMe, 25B-NBOMe, 25C-NBOMe, 25TFM-NBOMe, 2CBCB-NBOMe, 25CN-NBOH, 2CBFly-NBOMe)
- 2Cs (e.g., 2C-B, 2C-E, 2C-I, 2C-T-2, 2C-T-7, 2C-T-21)
- 2C-B-FLY
- 2CB-Ind
- 5-Methoxytryptamines (5-MeO-DET, 5-MeO-DiPT, 5-MeO-DMT, 5-MeO-DPT, 5-MT)
- α-Alkyltryptamines (e.g., 5-Cl-αMT, 5-Fl-αMT, 5-MeO-αET, 5-MeO-αMT, α-Me-5-HT, αET, αMT)
- AL-34662
- AL-37350A
- Bromo-DragonFLY
- Dimemebfe
- DMBMPP
- DOx (e.g., DOB, DOC, DOI, DOM)
- Efavirenz
- Ergolines (e.g., 1P-LSD, ALD-52, bromocriptine, cabergoline, ergine (LSA), ergotamine, lisuride, LA-SS-Az, LSB, LSD, LSD-Pip, LSH, LSP, methylergometrine (methylergonovine), pergolide)
- Flumexadol
- Jimscaline
- Lorcaserin
- MDxx (e.g., MDA, MDMA, MDOH, MMDA)
- O-4310
- Oxaflozane
- PHA-57378
- PNU-22394
- PNU-181731
- RH-34
- Phenethylamines (e.g., lophophine, mescaline)
- Piperazines (e.g., BZP, mCPP, quipazine, TFMPP)
- Serotonin (5-HT)
- TCB-2
- TFMFly
- Tryptamines (e.g., 5-BT, 5-CT, bufotenin, DET, DiPT, DMT, DPT, psilocin, psilocybin, tryptamine)
| |
- Antagonists: 5-I-R91150
- 5-MeO-NBpBrT
- AC-90179
- Adatanserin
- Altanserin
- AMDA
- APD-215
- Atypical antipsychotics (e.g., amperozide, aripiprazole, asenapine, blonanserin, carpipramine, clocapramine, clorotepine, clozapine, fluperlapine, gevotroline, iloperidone, melperone, mosapramine, ocaperidone, olanzapine, paliperidone, quetiapine, risperidone, sertindole, zicronapine, ziprasidone, zotepine)
- Cinanserin
- CSP-2503
- Cyproheptadine
- Deramciclane
- Dotarizine
- Eplivanserin
- Ergolines (e.g., amesergide, LY-53857, LY-215840, mesulergine, metergoline, methysergide, sergolexole)
- Etoperidone
- Fananserin
- Flibanserin
- Glemanserin
- Irindalone
- Ketanserin
- KML-010
- Lubazodone
- LY-393558
- Medifoxamine
- Mepiprazole
- Metitepine (methiothepin)
- MIN-101
- Naftidrofuryl
- Nantenine
- Nefazodone
- Pelanserin
- Phenoxybenzamine
- Pimavanserin
- Pirenperone
- Pizotifen
- Pruvanserin
- Rauwolscine
- Ritanserin
- S-14671
- Sarpogrelate
- Setoperone
- Spiperone
- Spiramide
- SR-46349B
- TGBA01AD
- Teniloxazine
- Temanogrel
- Tetracyclic antidepressants (e.g., amoxapine, aptazapine, esmirtazapine, maprotiline, mianserin, mirtazapine)
- Trazodone
- Tricyclic antidepressants (e.g., amitriptyline)
- Typical antipsychotics (e.g., chlorpromazine, fluphenazine, haloperidol, loxapine, perphenazine, pimozide, pipamperone, prochlorperazine, thioridazine, thiothixene, trifluoperazine)
- Volinanserin
- Xylamidine
- Yohimbine
| | |
|
|---|
| | 5-HT2B |
- Agonists: 4-Methylaminorex
- Aminorex
- Amphetamines (eg., chlorphentermine, cloforex, dexfenfluramine, fenfluramine, levofenfluramine, norfenfluramine)
- BW-723C86
- DOx (e.g., DOB, DOC, DOI, DOM)
- Ergolines (e.g., cabergoline, dihydroergocryptine, dihydroergotamine, ergotamine, methylergometrine (methylergonovine), methysergide, pergolide)
- MDxx (e.g., MDA, MDMA, MDOH, MMDA)
- Piperazines (e.g., mCPP)
- PNU-22394
- Ro60-0175
- Serotonin (5-HT)
- Tryptamines (e.g., 5-BT, 5-CT, 5-MT, α-Me-5-HT, bufotenin, DET, DiPT, DMT, DPT, psilocin, psilocybin, tryptamine)
| | | | |
|
|---|
| | 5-HT2C |
- Agonists: 2Cs (e.g., 2C-B, 2C-E, 2C-I, 2C-T-2, 2C-T-7, 2C-T-21)
- 5-Methoxytryptamines (5-MeO-DET, 5-MeO-DiPT, 5-MeO-DMT, 5-MeO-DPT, 5-MT)
- α-Alkyltryptamines (e.g., 5-Cl-αMT, 5-Fl-αMT, 5-MeO-αET, 5-MeO-αMT, α-Me-5-HT, αET, αMT)
- A-372159
- AL-38022A
- Alstonine
- CP-809101
- Dimemebfe
- DOx (e.g., DOB, DOC, DOI, DOM)
- Ergolines (e.g., ALD-52, cabergoline, dihydroergotamine, ergine (LSA), ergotamine, lisuride, LA-SS-Az, LSB, LSD, LSD-Pip, LSH, LSP, pergolide)
- Flumexadol
- Lorcaserin
- MDxx (e.g., MDA, MDMA, MDOH, MMDA)
- MK-212
- Org 12962
- Org 37684
- Oxaflozane
- PHA-57378
- Phenethylamines (e.g., lophophine, mescaline)
- Piperazines (e.g., aripiprazole, BZP, mCPP, quipazine, TFMPP)
- PNU-22394
- PNU-181731
- Ro60-0175
- Ro60-0213
- Serotonin (5-HT)
- Tryptamines (e.g., 5-BT, 5-CT, bufotenin, DET, DiPT, DMT, DPT, psilocin, psilocybin, tryptamine)
- Vabicaserin
- WAY-629
- WAY-161503
- YM-348
| |
- Antagonists: Adatanserin
- Agomelatine
- Atypical antipsychotics (e.g., asenapine, clorotepine, clozapine, fluperlapine, iloperidone, melperone, olanzapine, paliperidone, quetiapine, risperidone, sertindole, ziprasidone, zotepine)
- Captodiame
- CEPC
- Cinanserin
- Cyproheptadine
- Deramciclane
- Dotarizine
- Eltoprazine
- Ergolines (e.g., amesergide, bromocriptine, LY-53857, LY-215840, mesulergine, metergoline, methysergide, sergolexole)
- Etoperidone
- Fluoxetine
- FR-260010
- Irindalone
- Ketanserin
- Ketotifen
- Latrepirdine (dimebolin)
- Medifoxamine
- Metitepine (methiothepin)
- Nefazodone
- Pirenperone
- Pizotifen
- Propranolol
- Ritanserin
- RS-102221
- S-14671
- SB-200646
- SB-206553
- SB-221284
- SB-228357
- SB-242084
- SB-243213
- SDZ SER-082
- Tedatioxetine
- Tetracyclic antidepressants (e.g., amoxapine, aptazapine, esmirtazapine, maprotiline, mianserin, mirtazapine)
- TIK-301
- Trazodone
- Tricyclic antidepressants (e.g., amitriptyline, nortriptyline)
- Typical antipsychotics (e.g., chlorpromazine, loxapine, pimozide, pipamperone, thioridazine)
- Xylamidine
| | |
|
|---|
|
|
|
| |
|---|
| | 5-HT3 |
- Agonists: Alcohols (e.g., butanol, ethanol, trichloroethanol)
- m-CPBG
- Phenylbiguanide
- Piperazines (e.g., BZP, mCPP, quipazine)
- RS-56812
- Serotonin (5-HT)
- SR-57227
- SR-57227A
- Tryptamines (e.g., 2-Me-5-HT, 5-CT, bufotenidine (5-HTQ))
- Volatiles/gases (e.g., halothane, isoflurane, toluene, trichloroethane)
- YM-31636
| |
- Antagonists: Alosetron
- AS-8112
- Atypical antipsychotics (e.g., clozapine, olanzapine, quetiapine)
- Azasetron
- Batanopride
- Bemesetron (MDL-72222)
- Cilansetron
- CSP-2503
- Dazopride
- Dolasetron
- Galanolactone
- Granisetron
- ICS-205930
- Lerisetron
- Memantine
- Ondansetron
- Palonosetron
- Ramosetron
- Renzapride
- Ricasetron
- Tedatioxetine
- Tetracyclic antidepressants (e.g., amoxapine, mianserin, mirtazapine)
- Thujone
- Tropanserin
- Tropisetron
- Typical antipsychotics (e.g., loxapine)
- Volatiles/gases (e.g., nitrous oxide, sevoflurane, xenon)
- Vortioxetine
- Zacopride
- Zatosetron
| | |
|
|---|
| | 5-HT4 | |
|---|
| | 5-HT5A | |
|---|
| | 5-HT6 |
- Agonists: Ergolines (e.g., dihydroergocryptine, dihydroergotamine, ergotamine, lisuride, LSD, mesulergine, metergoline, methysergide)
- Serotonin (5-HT)
- Tryptamines (e.g., 2-Me-5-HT, 5-BT, 5-CT, 5-MT, Bufotenin, E-6801, E-6837, EMD-386088, EMDT, LY-586713, N-Me-5-HT, tryptamine)
- WAY-181187
- WAY-208466
| |
- Antagonists: ABT-354
- Atypical antipsychotics (e.g., aripiprazole, asenapine, clorotepine, clozapine, fluperlapine, iloperidone, olanzapine, tiospirone)
- AVN-101
- AVN-211
- AVN-322
- AVN-397
- BGC20-760
- BVT-5182
- BVT-74316
- Cerlapirdine
- EGIS-12233
- GW-742457
- Idalopirdine
- Ketanserin
- Latrepirdine (dimebolin)
- Metitepine (methiothepin)
- MS-245
- PRX-07034
- Ritanserin
- Ro04-6790
- Ro 63-0563
- SB-258585
- SB-271046
- SB-357134
- SB-399885
- SB-742457
- Tetracyclic antidepressants (e.g., amoxapine, mianserin)
- Tricyclic antidepressants (e.g., amitriptyline, clomipramine, doxepin, nortriptyline)
- Typical antipsychotics (e.g., chlorpromazine, loxapine)
| | |
|
|---|
| | 5-HT7 | | |
- Antagonists: Atypical antipsychotics (e.g., amisulpride, aripiprazole, asenapine, clorotepine, clozapine, fluperlapine, olanzapine, risperidone, sertindole, tiospirone, ziprasidone, zotepine)
- Butaclamol
- DR-4485
- EGIS-12233
- Ergolines (e.g., 2-Br-LSD (BOL-148), amesergide, bromocriptine, cabergoline, dihydroergotamine, ergotamine, LY-53857, LY-215840, mesulergine, metergoline, methysergide, sergolexole)
- JNJ-18038683
- Ketanserin
- LY-215840
- Metitepine (methiothepin)
- Ritanserin
- SB-258719
- SB-258741
- SB-269970
- SB-656104
- SB-656104A
- SB-691673
- SLV-313
- SLV-314
- Spiperone
- SSR-181507
- Tetracyclic antidepressants (e.g., amoxapine, maprotiline, mianserin, mirtazapine)
- Tricyclic antidepressants (e.g., amitriptyline, clomipramine, imipramine)
- Typical antipsychotics (e.g., acetophenazine, chlorpromazine, chlorprothixene, fluphenazine, loxapine, pimozide)
- Vortioxetine
| | |
|
|---|
|
|
|
- See also: Adrenergics
- Dopaminergics
- Melatonergics
- Monoamine reuptake and release modulators
- Monoamine metabolism modulators
- Monoamine neurotoxins
|

 . PMID 13898151.
. PMID 13898151.