Irinotecan
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Camptosar, Campto |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608043 |
| Pregnancy category |
|
| Routes of administration | Intravenous |
| ATC code | L01XX19 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | NA |
| Metabolism | Hepatic glucuronidation |
| Biological half-life | 6 to 12 hours |
| Excretion | Biliary and renal |
| Identifiers | |
| |
| CAS Number |
97682-44-5 |
| PubChem (CID) | 60838 |
| IUPHAR/BPS | 6823 |
| DrugBank |
DB00762 |
| ChemSpider |
54825 |
| UNII |
7673326042 |
| KEGG |
D08086 |
| ChEBI |
CHEBI:80630 |
| ChEMBL |
CHEMBL481 |
| ECHA InfoCard | 100.127.784 |
| Chemical and physical data | |
| Formula | C33H38N4O6 |
| Molar mass |
586.678 g/mol (Irinotecan) 623.139 g/mol (Irinotecan hydrochloride) 677.185 g/mol (Irinotecan hydrochloride trihydrate) |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Irinotecan, sold under the brand name Camptosar, is a medication used for the treatment of cancer. Its main use is in colon cancer, in particular, in combination with other chemotherapy agents.
Irinotecan prevents DNA from unwinding by inhibition of topoisomerase 1.[1] In chemical terms, it is a semisynthetic molecule similar to the natural alkaloid camptothecin.
It is on the WHO Model List of Essential Medicines, the most important medications needed in a basic health system.[2] Irinotecan received accelerated approval from the U.S. Food and Drug Administration (FDA) in 1996 and full approval in 1998.[3][4]
Medical uses
Its main use is in colon cancer, in particular, in combination with other chemotherapy agents. This includes the regimen FOLFIRI, which consists of infusional 5-fluorouracil, leucovorin, and irinotecan.
Side-effects
The most significant adverse effects of irinotecan are severe diarrhea and extreme suppression of the immune system.
Diarrhea
Irinotecan-associated diarrhea is severe and clinically significant, sometimes leading to severe dehydration requiring hospitalization or intensive care unit admission. This side-effect is managed with the aggressive use of antidiarrheals such as loperamide or co-phenotrope with the first loose bowel movement.
Immunosuppression
The immune system is adversely impacted by irinotecan. This is reflected in dramatically lowered white blood cell counts in the blood, in particular the neutrophils. The patient may experience a period of neutropenia (a clinically significant decrease of neutrophils in the blood) while the bone marrow increases white cell production to compensate.
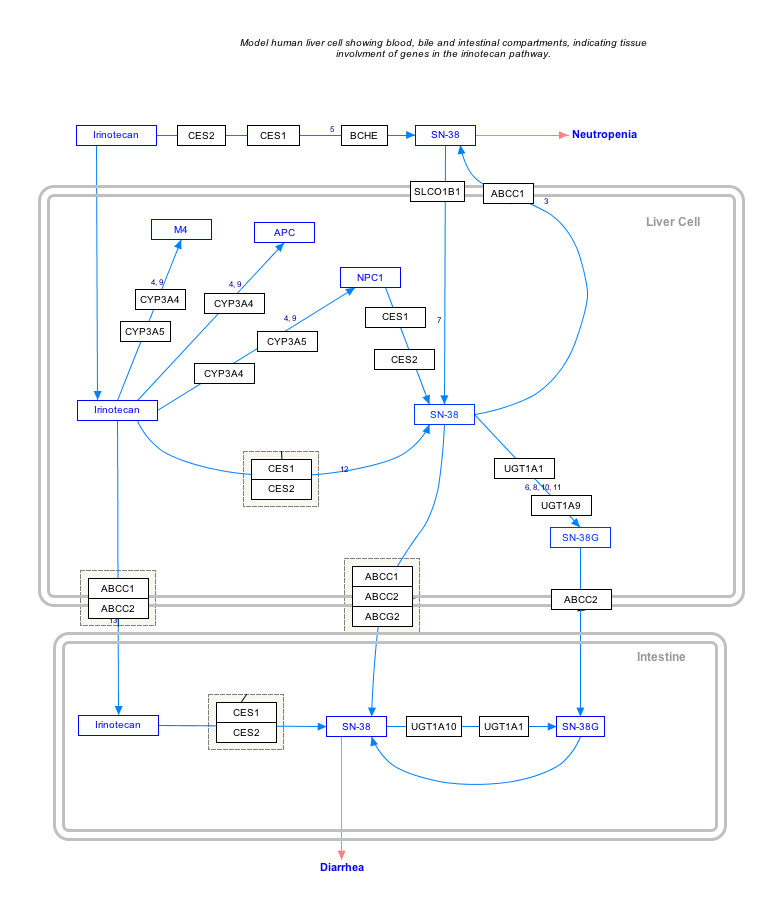
Mechanism
Irinotecan is activated by hydrolysis to SN-38, an inhibitor of topoisomerase I. This is then inactivated by glucuronidation by uridine diphosphate glucoronosyltransferase 1A1 (UGT1A1). The inhibition of topoisomerase I by the active metabolite SN-38 eventually leads to inhibition of both DNA replication and transcription.
The molecular action of irinotecan occurs by trapping a subset of topoisomerase-1-DNA cleavage complexes, those with a guanine +1 in the DNA sequence.[5] One irinotecan molecule stacks against the base pairs flanking the topoisomerase-induced cleavage site and poisons (inactivates) the topoisomerase 1 enzyme.[5]
Interactive pathway
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
Irinotecan Pathway edit
- ↑ The interactive pathway map can be edited at WikiPathways: "IrinotecanPathway_WP46359".
Pharmacogenomics
Irinotecan is converted by an enzyme into its active metabolite SN-38, which is in turn inactivated by the enzyme UGT1A1 by glucuronidation.
*28 variant patients
People with variants of the UGT1A1 called TA7, also known as the "*28 variant", express fewer UGT1A1 enzymes in their liver and often have Gilbert's syndrome. During chemotherapy, they effectively receive a larger than expected dose because their bodies are not able to clear irinotecan as fast as others. In studies this corresponds to higher incidences of severe neutropenia and diarrhea.[6]
In 2004, a clinical study was performed that both validated prospectively the association of the *28 variant with greater toxicity and the ability of genetic testing in predicting that toxicity before chemotherapy administration.[6]
In 2005, the FDA made changes to the labeling of irinotecan to add pharmacogenomics recommendations, such that irinotecan recipients with a homozygous (both of the two gene copies) polymorphism in UGT1A1 gene, to be specific, the *28 variant, should be considered for reduced drug doses.[7] Irinotecan is one of the first widely used chemotherapy agents that is dosed according to the recipient's genotype.[8]
Research
In 2014, it was shown that antitumor activity of irinotecan against glioblastoma can be enhanced by co-treatment with statins.[9] Similarly, it was shown that berberine may enhance chemosensitivity to irinotecan in colon cancer cells.[10]
Formulations
Liposome encapsulated
A liposome encapsulated version of irinotecan sold as Onivyde, was approved by FDA in October 2015 to treat metastatic pancreatic cancer.[11] It gained EU approval in October 2016.[12]
Names
During development, it was known as CPT-11.
References
- ↑ Pommier, Y.; Leo, E.; Zhang, H.; Marchand, C. (2010). "DNA topoisomerases and their poisoning by anticancer and antibacterial drugs". Chem. Biol. 17: 421–433.
- ↑ "19th WHO Model List of Essential Medicines (April 2015)" (PDF). WHO. April 2015. Retrieved May 10, 2015.
- ↑ New York Times Article http://www.nytimes.com/1996/06/18/science/new-cancer-drug-approved.html
- ↑ FDA Review Letter http://www.accessdata.fda.gov/drugsatfda_docs/appletter/1998/20571s8ltr.pdf
- 1 2 Pommier Y (2013). "Drugging topoisomerases: lessons and challenges". ACS Chem. Biol. 8 (1): 82–95. doi:10.1021/cb300648v. PMC 3549721
 . PMID 23259582.
. PMID 23259582. - 1 2 Innocenti F, Undevia SD, Iyer L, et al. (April 2004). "Genetic variants in the UDP-glucuronosyltransferase 1A1 gene predict the risk of severe neutropenia of irinotecan". J. Clin. Oncol. 22 (8): 1382–8. doi:10.1200/JCO.2004.07.173. PMID 15007088.
- ↑ Camptosar® irinotecan hydrochloride injection August 2010 http://labeling.pfizer.com/ShowLabeling.aspx?id=533
- ↑ O'Dwyer PJ, Catalano RB (October 2006). "Uridine diphosphate glucuronosyltransferase (UGT) 1A1 and irinotecan: practical pharmacogenomics arrives in cancer therapy". J. Clin. Oncol. 24 (28): 4534–8. doi:10.1200/JCO.2006.07.3031. PMID 17008691.
- ↑ Jiang PF (Jan 2014). "Novel anti-glioblastoma agents and therapeutic combinations identified from a collection of FDA approved drugs.". J Transl Med. 12. doi:10.1186/1479-5876-12-13. PMC 3898565
 . PMID 24433351.
. PMID 24433351. - ↑ Yu M (Jan 2014). "Berberine enhances chemosensitivity to irinotecan in colon cancer via inhibition of NF-κB". J Mol Med Rep. 9 (1): 249–54. doi:10.3892/mmr.2013.1762. PMID 24173769.
- ↑ News Release (22 October 2015). "FDA approves new treatment for advanced pancreatic cancer". FDA.
- ↑ Shire’s leading Pancreatic Cancer drug is Back with a Bang. 2016