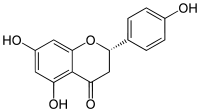
Naringenin
 | |
| Names | |
|---|---|
| IUPAC name
5,7-Dihydroxy-2-(4-hydroxyphenyl)chroman-4-one | |
| Other names
Naringetol; Salipurol; Salipurpol; 4',5,7-Trihydroxyflavanone | |
| Identifiers | |
| 480-41-1 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:50202 |
| ChEMBL | ChEMBL9352 |
| ChemSpider | 388383 |
| DrugBank | DB03467 |
| ECHA InfoCard | 100.006.865 |
| PubChem | 439246 |
| UNII | HN5425SBF2 |
| |
| |
| Properties | |
| C15H12O5 | |
| Molar mass | 272.26 g·mol−1 |
| Melting point | 251 °C (484 °F; 524 K)[1] |
| 475 mg/L[1] | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Naringenin is a flavanone, a type of flavonoid. It is the predominant flavanone in grapefruit.[2]
Sources and bioavailability
Naringenin can be found in grapefruit, oranges, tomatoes (skin)[3] and in water mint.
This bioflavonoid is difficult to absorb on oral ingestion. In the best-case scenario, only 15% of ingested naringenin will get absorbed in the human gastrointestinal tract.
The naringenin-7-glucoside form seems less bioavailable than the aglycol form.[4]
Grapefruit juice can provide much higher plasma concentrations of naringenin than orange juice.[5] Also found in grapefruit is the related compound kaempferol, which has a hydroxyl group next to the ketone group.
Naringenin can be absorbed from cooked tomato paste.[6]
Potential biological effects
Naringenin has been shown to have an inhibitory effect on the human cytochrome P450 isoform CYP1A2, which can change pharmacokinetics in a human (or orthologous) host of several popular drugs in an adverse manner, even resulting in carcinogens of otherwise harmless substances.[7] The National Research Institute of Chinese Medicine in Taiwan conducted experiments on the effects of the grapefruit flavanones naringin and naringenin on CYP450 enzyme expression. Naringenin proved to be a potent inhibitor of the benzo(a)pyrene metabolizing enzyme benzo(a)pyrene hydroxylase (AHH) in experiments in mice.[8]
Naringenin has also been shown to reduce oxidative damage to DNA in vitro and in animal studies.[9]
Naringenin has also been shown to reduce hepatitis C virus production by infected hepatocytes (liver cells) in cell culture. This seems to be secondary to naringenin's ability to inhibit the secretion of very-low-density lipoprotein by the cells.[10] The antiviral effects of naringenin are currently under clinical investigation.[11]
Naringenin seems to protect LDLR-deficient mice from the obesity effects of a high-fat diet.[12]
Naringenin lowers the plasma and hepatic cholesterol concentrations by suppressing HMG-CoA reductase and ACAT in rats fed a high-cholesterol diet.[13]
It also produces BDNF-dependent antidepressant-like effects in mice.[14]
Like many other flavonoids, naringenin has been found to possess weak activity at the opioid receptors.[15] It specifically acts as a non-selective antagonist of all three opioid receptors, albeit with weak affinity.[15]
Metabolism
The enzyme naringenin 8-dimethylallyltransferase uses dimethylallyl diphosphate and (−)-(2S)-naringenin to produce diphosphate and 8-prenylnaringenin.
Biodegradation
Cunninghamella elegans, a fungal model organism of the mammalian metabolism, can be used to study the naringenin sulfation.[16]
References
- 1 2 "Naringenin". ChemIDplus.
- ↑ Felgines C, Texier O, Morand C, Manach C, Scalbert A, Régerat F, Rémésy C (December 2000). "Bioavailability of the flavanone naringenin and its glycosides in rats". Am. J. Physiol. Gastrointest. Liver Physiol. 279 (6): G1148–54. PMID 11093936.
- ↑ Vallverdú-Queralt, A; Odriozola-Serrano, I; Oms-Oliu, G; Lamuela-Raventós, RM; Elez-Martínez, P; Martín-Belloso, O (2012). "Changes in the polyphenol profile of tomato juices processed by pulsed electric fields". J Agric Food Chem. 60 (38): 9667–9672. doi:10.1021/jf302791k. PMID 22957841.
- ↑ Choudhury R, Chowrimootoo G, Srai K, Debnam E, Rice-Evans CA (November 1999). "Interactions of the flavonoid naringenin in the gastrointestinal tract and the influence of glycosylation". Biochem. Biophys. Res. Commun. 265 (2): 410–5. doi:10.1006/bbrc.1999.1695. PMID 10558881.
- ↑ Erlund I, Meririnne E, Alfthan G, Aro A (February 2001). "Plasma kinetics and urinary excretion of the flavanones naringenin and hesperetin in humans after ingestion of orange juice and grapefruit juice". J. Nutr. 131 (2): 235–41. PMID 11160539.
- ↑ Bugianesi R, Catasta G, Spigno P, D'Uva A, Maiani G (November 2002). "Naringenin from cooked tomato paste is bioavailable in men". J. Nutr. 132 (11): 3349–52. PMID 12421849.
- ↑ Fuhr U, Klittich K, Staib AH (April 1993). "Inhibitory effect of grapefruit juice and its bitter principal, naringenin, on CYP1A2 dependent metabolism of caffeine in man". Br J Clin Pharmacol. 35 (4): 431–6. doi:10.1016/0024-3205(96)00417-1. PMC 1381556
 . PMID 8485024.
. PMID 8485024. - ↑ Ueng YF, Chang YL, Oda Y, Park SS, Liao JF, Lin MF, Chen CF (1999). "In vitro and in vivo effects of naringin on cytochrome P450-dependent monooxygenase in mouse liver". Life Sci. 65 (24): 2591–602. doi:10.1016/s0024-3205(99)00528-7. PMID 10619367.
- ↑ Sumit Kumar & Ashu Bhan Tiku (2016). "Biochemical and Molecular Mechanisms of Radioprotective Effects of Naringenin, a Phytochemical from Citrus Fruits". J. Agric. Food Chem. 64 (8): 1676–1685. doi:10.1021/acs.jafc.5b05067.
- ↑ Nahmias Y, Goldwasser J, Casali M, van Poll D, Wakita T, Chung RT, Yarmush ML (May 2008). "Apolipoprotein B-dependent hepatitis C virus secretion is inhibited by the grapefruit flavonoid naringenin". Hepatology. 47 (5): 1437–45. doi:10.1002/hep.22197. PMID 18393287.
- ↑ A Pilot Study of the Grapefruit Flavonoid Naringenin for HCV Infection
- ↑ Mulvihill EE, Allister EM, Sutherland BG, Telford DE, Sawyez CG, Edwards JY, Markle JM, Hegele RA, Huff MW (October 2009). "Naringenin prevents dyslipidemia, apolipoprotein B overproduction, and hyperinsulinemia in LDL receptor-null mice with diet-induced insulin resistance". Diabetes. 58 (10): 2198–210. doi:10.2337/db09-0634. PMC 2750228
 . PMID 19592617.
. PMID 19592617. - ↑ Lee SH, Park YB, Bae KH, Bok SH, Kwon YK, Lee ES, Choi MS (1999). "Cholesterol-lowering activity of naringenin via inhibition of 3-hydroxy-3-methylglutaryl coenzyme A reductase and acyl coenzyme A:cholesterol acyltransferase in rats". Ann. Nutr. Metab. 43 (3): 173–80. doi:10.1159/000012783. PMID 10545673.
- ↑ Yi LT, Liu BB, Li J, Luo L, Liu Q, Geng D, Tang Y, Xia Y, Wu D (October 2013). "BDNF signaling is necessary for the antidepressant-like effect of naringenin". Prog. Neuropsychopharmacol. Biol. Psychiatry. 48C: 135–141. doi:10.1016/j.pnpbp.2013.10.002. PMID 24121063.
- 1 2 Katavic PL, Lamb K, Navarro H, Prisinzano TE (August 2007). "Flavonoids as opioid receptor ligands: identification and preliminary structure-activity relationships". J. Nat. Prod. 70 (8): 1278–82. doi:10.1021/np070194x. PMC 2265593
 . PMID 17685652.
. PMID 17685652. - ↑ Ibrahim AR (January 2000). "Sulfation of naringenin by Cunninghamella elegans". Phytochemistry. 53 (2): 209–12. doi:10.1016/S0031-9422(99)00487-2. PMID 10680173.